Safety, tolerability, and preliminary immunogenicity of a recombinant Staphylococcus aureus vaccine (RSAV) in healthy adults between 18 and 65 years in China: study protocol for a randomized, double-blind, placebo-controlled, phase 1 trial
Introduction
Staphylococcus aureus (S. aureus), a gram positive bacteria, is one of a major cause of hospital-associated and community-associated infections worldwide (1). S. aureus accounted for 22% of all blood infections (8,929 of 40,497 infections), 23.2% of all lower respiratory tract (3,371/14,552 infections) and 39.2% (2,928/7,474 infections) of all skin and soft tissue infections, in the USA, Canada, and Europe. In China, S. aureus was the most common clinical gram-positive bacteria with the highest isolated rate, and the Methicillin-resistant S. aureus (MRSA) accounted for 44.6% of all the isolated strains (3). The frequencies of both community-acquired and hospital-acquired staphylococcal infections have increased steadily in recent years. Treatment of these infections caused by S. aureus has become more difficult because of the emergence of multidrug-resistant strains (4). It is critical to develop an efficacy vaccine to control the infection caused by S. aureus and reduce the usage of antibiotics in order to decrease the drug resistance of S. aureus.
Up to now, several S. aureus vaccine candidates had been evaluated in clinical trials, including StaphVax developed by Nabi Company (the U.S.), and a recombinant subunit vaccine V710 developed by Merck. Even though both candidates induced a strong immune response in the recipients in the early clinical trials, no effective protection against the S. aureus infections was found in the phase 3 efficacy trials (5,6). Currently, several clinical trials with other novel S. aureus vaccine candidates are still ongoing, the protective effects of the vaccines against S. aureus infection has not been proved yet (7).
Recently, a novel a recombinant Staphylococcus aureus vaccine (RSAV) has been developed in China, which showed a good safety and immunogenicity profile in the preclinical studies. We reported the protocol of this phase 1 clinical trial of the RSAV. The trial aims to evaluate the safety, tolerability and the preliminary immunogenicity of the RSAV in healthy adults.
Methods
Study design and population
This study is a single-center, double-blind, placebo-controlled clinical trial of a novel RSAV in Taixing, Jiangsu Province, China, in September 2016. Healthy participants aged between 18 and 65 years will be recruited. Written informed consent will be obtained from each participant before any study-related activity began. Routine blood, blood biochemical and axillary temperature will be performed at the enrollment for selecting participants. Eligible participants should be physically healthy, with an axillary temperature no more than 37 °C at the enrollment, have no prior previous S. aureus infection in the last 12 months, immunization with other S. aureus vaccine candidates. Exclusion criteria are pregnancy, positive HIV, history of autoimmune disorders, and so on. The Table 1 provides a complete list of inclusion and exclusion criteria. The trial was approved by the Ethics Committee of Jiangsu Provincial Center for Disease Prevention and Control and will be performed in compliance with the Declaration of Helsinki and Good Clinical Practice. The trial was registered with a ClinicalTrials.gov Identifier: NCT02804711. The full trial protocol of this trial is available with the Appendix.
Table 1
| Inclusion criteria |
| Healthy volunteers aged 18 to 65 years (aged over 18 and under 66 years) |
| Able to comply with all clinical trial protocol requirements and willing to complete all the visit plan process during the whole clinical trial observation period |
| Able to understand the content of informed consent and willing to sign the informed consent |
| Able to complete the diary card independently |
| For females only (18–49 years), a negative urine pregnancy test and willing to practice continuous effective contraception during the study |
| Axillary temperature ≤37.0 °C |
| Exclusion criteria |
| First Immunization exclusion standard: |
| Prior receipt of Staphylococcus aureus vaccine |
| Any confirmed Staphylococcus aureus infection disease in the past 12 months |
| History of asthma, hereditary angioneurotic edema, diabetes, malignancy or other serious disease. Participation in the clinical trial is likely to increase the disease risk and interfere with the observation of clinical trial |
| Prior blood donation or blood loss over 400 mL in the last 3 months |
| Coagulation disorders (coagulation factor deficiency, coagulopathy or platelet disorder) diagnosed by doctors, or obvious bruises or blood coagulation noticed |
| History of allergic disease likely to be exacerbated by any component of the vaccine, including allergy, urticaria, respiratory difficulty, angioneurotic edema or abdominal pain |
| Any autoimmune disease or immunodeficient state, parents, brother and sister with autoimmune disease or immunodeficient disease |
| Taking immunoglobulins and/or any blood products within the last 12 months |
| Asplenia, functional asplenia or asplenia caused by any situation or splenectomy |
| Any acute disease or acute attack of chronic disease in last 7 days |
| History of thyroidectomy or thyroid disease requiring treatment in the last 12 months |
| Immunosuppressor, cytotoxic therapy, inhaled corticosteroid (excluding corticosteroids spray treatment of allergic rhinitis, acute and non-concurrent corticosteroids treatment) |
| Participation in another research study involving receipt of an investigational product in the last 30 days |
| Woman who is breast-feeding |
| Prior administration of attenuated vaccine in last 28 days |
| Prior administration of subunit vaccine, inactivated vaccine or allergic therapy in last 14 days |
| Current anti-tuberculosis prophylaxis or therapy |
| Any other conditions may compromise the safety or availability of participants in the judgment of the investigator |
| Following immunization exclusion standard: |
| Any grade 3 or more serious adverse reaction happen since the last vaccination |
| Other condition violates the inclusion criteria or meets the exclusion criteria is noticed after the first immunization |
| Acute or chronic infections at the vaccination day (axillary temperature >37.0 °C) |
Randomization allocation and blinding
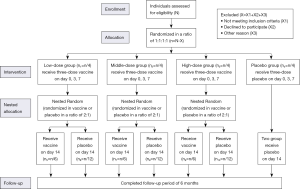
A total of 144 participants will be included and then randomly allocated in a ratio of 1:1:1:1 to the low-dose group, middle-dose group, high-dose group, or placebo group for the first three-dose vaccination. A nested randomization is performed for the fourth dose vaccination: participants within each treatment group will be randomly assigned in a ratio of 2:1 to receive a fourth shot of vaccine or placebo (Figure 1). All participants, investigators and study staffs participating in this study will be masked to the treatment allocation.
The investigational vaccines and placebo will be randomized and coded according to a randomization list generated by an independent statistician using SAS (version, 9.3). All the randomized vaccine and placebo doses will be identical in the appearance, with labeled randomized numbers as sole identifier for each dose. The person who involved in randomizing and blinding shall not participate in any other process of the trial, also shall not disclose the contents of blinding to any personnel to participate in clinical trial work.
Intervention
Experimental vaccine RSAV is co-developed by Chengdu Olymvax Biopharmaceuticals Inc. and Third Military Medical University. Vaccine contains HI, SpA5, mSEB, MntC recombinant proteins and aluminum phosphate adjuvants of 1.952 mg per dose. HI is a fusion protein combined by two antigens IsdB and HLa. The final product is 0.6 mL mixed suspension per vial for injection. The low dose, medium dose, high dose contain 15, 30, and 60 µg, for each protein per dose, respectively. The placebo contained the vaccine excipients only, with aluminum phosphate adjuvants of 1.952 mg and no proteins per dose.
Procedure
The injections will be given intramuscularly in the deltoid muscle in participants on day 0, day 3, day 7, day 14. We will observe participants for immediate adverse reactions for 30 minutes after vaccination at the clinic site. Participants will be followed for 21 days post-vaccination to assess any solicited injection-site or systemic adverse reactions and any unsolicited adverse events or medication use for 42 days post-vaccination. Serious adverse events occurred during the whole study period will be documented. Blood samples will be collected at baseline prior to vaccination and at each programmed visit after vaccination. Nose swabs will be collected from participants follow-up visits at day 3, 7, 10, 14, 17, 21, 42, 3 months and 6 months after injection. A total of 11 visits will be attended by participants, including the screening visit before the vaccination and 10 site visits during a follow-up period of 6 months (Table 2). Participants will be required to keep a record of diary between visits for the documentation of local or systemic adverse events. At every visit, diaries will be reviewed by a physician to verify the accuracy of reported symptoms, and participants will be interviewed for adverse events.
Table 2
| Activities | V0 | V1 | V2 | V3 | V4 | V5 | V6 | V7 | V8 | V9 | V10 | |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Within 7 days before day 0 | D 0 | D 3 | D 7 | D 10 | D 14 | D 17 | D 21 | D 42 | M 3 | M 6 | ||
| 1 | Informed consent | ☆ | ||||||||||
| 2 | Collecting demographic information | ☆ | ||||||||||
| 3 | Preliminarily check inclusion and exclusion criteria | ☆ | ||||||||||
| 4 | Physical examination | |||||||||||
| Height, weight, BMI | ☆ | |||||||||||
| Respiratory rate, pulse rate, blood pressure | ☆ | |||||||||||
| Urine pregnancy test (female) | ☆ | |||||||||||
| Routine blood test | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ||||||
| Blood biochemical test | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ||||||
| Pre-vaccination body temperature | ☆ | ☆ | ☆ | ☆ | ||||||||
| 5 | Inclusion and exclusion criteria before enrolled | ☆ | ||||||||||
| 6 | Follow-up vaccination exclusion criteria | ☆ | ☆ | ☆ | ||||||||
| 7 | Randomization | ☆ | ||||||||||
| 8 | Serum analyses: | |||||||||||
| Five antigen-specific antibody response by ELISA | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |||||
| Functional antibody level | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |||||
| Cellular immune responses | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |||||
| 9 | Nasal sampling swab | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |
| 10 | Vaccination | ☆ | ☆ | ☆ | ☆ | |||||||
| 11 | Observation for 30 minutes post-vaccination, participates safety observation training | ☆ | ☆ | ☆ | ☆ | |||||||
| 12 | Distribution of diary cards, ruler, signing-pen, thermometer | ☆ | ☆ | ☆ | ||||||||
| 13 | Record on the “Vaccination and visits record” | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |
| 14 | Participates safety observations self-report in diary cards | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |||
| 15 | Report serious adverse events | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |
| 16 | Return of diary cards | ☆ | ☆ | ☆ | ||||||||
Outcome measurement
The primary study objective is safety and tolerability of RSAV, the main safety outcome was measured with the incidence of solicited adverse reactions within 21 days. Change from baseline for safety laboratory measures (Routine blood and blood biochemical indexes) on day 3, day 7, day 10, day 14, day 17 are the secondary end-point variable of the study. Unsolicited adverse events within 42 days after vaccination and any occurrence of serious adverse events (SAE) during the six-month follow-up period will also be analyzed. Immunological response and vaccination efficacy are based on the following criteria: geometric mean titers (GMT), geometric mean fold increase (GMFI), seroconversion rate of antigen-specific antibodies in serum.
In order to study in-depth the immune response to RSAV in participants, we will also explore other biological outcomes in the collected samples. Cellular immune response to vaccination will be determined by Elispot on day 0 (immediately before vaccination), day 7, day 14, day 21, day 42, month 3 and month 6 after S. aureus vaccination. Functional antibody level detection will also be detected at day 0 (immediately before vaccination), day 7, day 14, day 21, day 42, month 3 and month 6. And the functional antibody level at different time will be compared to reveal the difference. In addition, nasal sampling swab will be detected for S. aureus colonization at day 0, day 3, day 7, day10, day 14, day 17, day 21, day 42, month 3 and month 6.
Sample size
According to the Chinese guidelines for vaccine clinical trial, the sample size of a phase 1 clinical trial should be around 20–30 (8). We do not calculate the sample size specifically according to statistical power. A total of 144 participants will be enrolled. Among them, 36 will receive the low dose vaccine, 36 will receive the high dose vaccine, 36 will receive the medium dose vaccine, and 36 will receive the placebo.
Data analysis
Safety analysis
All randomized participants who received vaccination should be included in the safety evaluation. Thus, the safety analysis will be performed on the basis of Intention-To-Treat (ITT) cohort. Safety analysis of this experiment is mainly descriptive analysis of incidence rate of adverse reaction or adverse events. A chi-square test can be used to compare the proportion of groups, Fisher’s exact test will be used when it is necessary. SAS (version 9.3) was used for all analyses, test statistics and the corresponding P values are given. All statistical tests were two-sided and significance was set at P≤0.05.
Immunogenicity analysis
Immunogenicity analysis will be performed using GMT, GMFI and seroconversion, we will compare vaccine groups versus placebo, in an ITT analysis using a logistic regression model with corresponding 95% confidence interval, and P value. Analysis of immunogenicity indicators on antibody levels need to do logarithmic transformation, the results of analysis should be shown in GMT, standard deviation, median, minimum and maximum values and 95% confidence intervals, chi-square test can be used to compare categorical indicators between groups such as positive conversion rate of immune response, if it is necessary, Fisher's exact test will be used. Statistical analysis method of repeated measures data can be used to analyze experimental data at different time points in this study.
Data and Safety Monitoring Board (DSMB)
All adverse events reported during the first 7 days after vaccination, grade 3 adverse experiences, serious adverse events report and new information probably affected adversely the safety of the participants or the conduct of the study will be reported to Data and Safety Monitoring Board (DSMB). DSMB will review all relative information of the serious adverse event and confirm it if it is vaccination related before unblinding. DSMB could make final decision to pause or all an early termination of the study. Administration of study injections and new enrollments will be paused according to the following situation: one serious adverse event may be associated with vaccination, or Occurrence of grade 3 adverse reaction may be associated with vaccination in 10% of participants or more (including injection-site reaction, systemic reaction, and change of the safety laboratory measures). Otherwise, The study may come to an early termination, if one vaccination-associated serious adverse event, or occurrence of grade 3 adverse events associated with vaccination in 15% of participants or more (including injection-site reaction, systemic reaction, and change of the safety laboratory measures),or required by sponsor, or required by regulatory authority, or required by institutional review board (IRB).
Ethical aspects and confidentiality of data
Investigators will obtain the written informed consent from each participant after providing them with appropriate information about the effects, objectives, methods, anticipated benefits and potential risks of participation. Investigators will also explain the right to withdraw consent at any time for any reason.
The trial staff ensured that participants’ confidentiality will be preserved. Prior to initiation of the trial, the investigators will sign a fully executed confidentiality agreement with the sponsor. All study-related information will be stored securely at the study sites and all participant information will be stored in locked file cabinets with access limited to study staff. All laboratory specimens, reports, study data collection, process, and administrative forms will be identified by coded number only to maintain participant confidentiality. Forms, lists, logbooks, appointment books, and any other listings that link participant numbers to other identifying information will be stored in a separate, locked file in an area with limited access.
Discussion
Before this trial, we conducted a first-in-human pilot study of the RSAV, which is a dose-escalated, open-label clinical trial. A total of 30 participants were enrolled and received two-dose vaccination of low-dose, middle-dose, or high-dose RSAV at day 0 and 7, respectively. Reactogenicity was closely monitored in these participants for 7 days after each dose, and the results supported us to continue evaluate the safety and immunogenicity of the RSAV this phase 1 trial (data has not been published).
There are several issues about the S. aureus vaccine need to be considered. Not like most preventative vaccines target on healthy people, the priority target populations of S. aureus vaccine were hospitalized patients undergoing invasive medical procedures such as surgery, or patients with chronic diseases such as those with end-stage renal disease (ESRD) and lived on hemodialysis. Generally, immunity systems of these patients probably were not as strong as those of healthy people. Unlike the normal vaccination regimen targeting on the healthy population, trying to elicit a strong and sustained immune response in the recipients. S. aureus vaccination may need to work as emergency vaccination, in order to quickly induce a protective response in the target populations to minimize the S. aureus infection risk, such as those going to have an operation. Clinical epidemiological data showed that the high-risk period of S. aureus infection is about 7 days after the injury or surgery (9). Therefore, we expected the intensive immunization regimens as three-dose at day 0, 3, 7 or four-dose at day 0, 3, 7, 14 could elicit a quick and strong protection. Thus, the dynamic change of antibody level against S. aureus in target populations should be followed closely.
This is the first study to evaluate the safety and immunogenicity profile of the RSAV in healthy adults. The results of this study would allow us to gain more insight in this novel S. aureus vaccine candidate and enable larger studies in the future.
Clinical trial contact list
Objectives and brief introduction
The recombinant staphylococcus aureus vaccine (RSAV) against Staphylococcus aureus (S. aureus) infection is co-developed by Chengdu Olymvax Biopharmaceuticals Inc. and Third Military Medical University. We are going to evaluate the safety, tolerability and immunogenicity of the novel RSAV in healthy adults aged between 18 and 65 years in China in this study.
In preclinical studies, the RSAV showed a good safety and immunogenicity profile. The RSAV has been approved by China Food and Drug Administration for clinical trials (approval number: 2015L01247). This protocol has been made according to Good Clinical Practice (GCP), the Declaration of Helsinki, and local rules and regulations of China.
Study site
This trial will be conducted by Jiangsu Provincial Center for Disease Control and Prevention. This trial is a single-center study, and the study site is in Taixing County, Jiangsu Province, China.
Background and rationale
Disease background
S. aureus, a gram positive bacteria, is one of a major cause of hospital-associated and community-associated infections worldwide. These infections caused by S. aureus ranged from minor skin and soft tissue infections to the major life-threatening invasive infections (1). The frequencies of both community-acquired and hospital-acquired S. aureus infections have increased steadily in recent years. Treatment of these infections caused by S. aureus has become more and more difficult because of the emergence of multidrug-resistant strains (4).
A comprehensive survey conducted in clinical sites in the USA, Canada, Europe, found that S. aureus accounted for 22% of all blood infections (8,929 of 40,497 infections), 23.2% of all lower respiratory tract (3,371/14,552 infections) and 39.2% (2,928/7,474 infections) of all skin and soft tissue infections (2). In China, S. aureus was the most common clinical gram-positive bacteria with the highest isolated rate, and the methicillin-resistant S. aureus (MRSA) accounted for 44.6% of all the isolated strains (3). It is critical to develop an effective vaccine to control the infection caused by S. aureus and reduce the usage of antibiotics in order to decrease the drug resistance of S. aureus.
Currently, there is no effective vaccine against S. aureus has been reported. Early clinical trials of several vaccine candidates such as Nabi’s StaphVax and PentaStaph, Intercell’s/Merck’s V710, VRi’s SA75 were all found to be invalid against infectious disease caused by S. aureus (7). Despite the fact that some vaccine candidates were highly immunogenic and able to elicit strong antibody responses, but no significant protection was noted. Moreover, some vaccine candidates even aggravated the risk of infection associated with S. aureus after vaccination (7). Up to now, none of these candidates was effective in preventing S. aureus infection.
The novel experimental vaccine under evaluation in this study, contains five antigens: the alpha hemolysin (Hla), iron-regulated surface determinant B (IsdB), S. aureus protein A (SpA), enterotoxin B (SEB), manganese transport protein C (MntC). In preclinical studies of this vaccine candidate, a total of 16,000 mice and 40 cynomolgus monkeys were tested. The vaccine was safe and immunogenic, showing a protection of over 85%. This is a phase 1 trial to evaluate the safety and tolerability and preliminary immunogenicity (including humoral immunity and cellular immunity) of this novel RSAV in healthy adults aged between 18–65 years.
Pathogeny characteristics
S. aureus could be typed through different methods including serological typing, phage typing, drug-resistant spectrum typing, genetic typing etc. The typing of S. aureus can be used to study the pathogenicity, drug resistance of S. aureus, and the relationship between the epidemiological characteristics and bacterial identification. According to the differences of enterotoxin antigenicity and isoelectric point, the S. aureus can be typed into A, B, C1, C2, C3, D, E, and F. Of them, S. aureus type A and B are most common. This bacteria has multiple antigens or cross-reactive antigens, which are poor in immunogenicity.
S. aureus as pathogenic bacteria widely exist in the environment. It can produce a variety of pathogenic toxin associated with various infectious diseases (4). Human are the main reservoir of S. aureus, and the bacteria is frequently colonized in the nose, respiratory tract, skin wounds, trachea incision site and even on the normal skin. The carriers of S. aureus have no symptom of infections, but are the important source of infection, causing nosocomial infection or community infection through nasal pharyngeal airway, skin wound, blood, or other routines. In addition, the S. aureus also can cause infection through contaminated food.
At hospital, population with the most high-risk of S. aureus infection are the surgery patients, ICU patients, long-term hemodialysis patients, large area burn patients, diabetes and organization transplantation patients. In community, children, adolescents, soldiers, and athletes have an injury of skin soft tissue and immunocompromised elderly people are also high risk group for the community-source S. aureus infection.
Vaccine development in other countries
Up to now, clinical trials of several S. aureus vaccines have been launched by biopharmaceutical companies, including Merck, Pfizer, Novartis, GlaxoSmithKline (GSK), The National Institute of Allergy and Infectious Diseases (NIAID) and so on (Table S1) (10).
Nabi Company (the U.S.) developed a novel capsular polysaccharide S. aureus vaccine StaphVax. The phase 3 clinical trials of StaphVax involved in 3,600 patients with hemodialysis and followed for S. aureus infection after vaccination from 3 to 35 weeks. Results showed that the StaphVax could not provide protection against S. aureus infection (5). In another phase 2 clinical trial for passive immunization of CP5 and CP8 antibodies which was isolated from healthy volunteers vaccinated with StaphVax (StaphVax indued a high level of CP5 and CP8 antibodies in serum), confirmed that CP5 and CP8 antibodies were not able to reduce the risk of S. aureus bacteremia (6). The results indicated that vaccine only contained S. aureus capsular polysaccharide antigen may not be able to provide protection against S. aureus infection.
Merck developed a recombinant subunit vaccine V710, which contains IsdB as antigen. IsdB is a conservative protein wildly existed in different isolates of MRSA, which is an anchor protein of cell wall and plays a key role in absorption of heme iron. In animal models, recombinant mice immunized with IsdB demonstrated a higher survival rate when exposed to lethal challenge with S. aureus. Besides, IsdB also showed a good immunogenicity in rhesus monkey. Moreover, a monoclonal antibody CS-D7 against IsdB showed a significant protection in mice challenging models. These results indicated that IsdB was a promising antigen for S. aureus vaccine. Based on above evidences, Merck cooperated with Intercell to develop the vaccine candidate V710 using IsdB as antigen. Between December 2007 and August 2011, a double-blind, randomized, event-driven trial in heart surgery patients was conducted at 165 sites in 26 countries. However, an interim analysis results showed that no significant difference between the vaccine and placebo recipients in terms of S. aureus infection incidence, even though V710 induced a strong immune response in the vaccine recipients (9). V710 could not only be able to reduce the rate of serious postoperative S. aureus infections among the patients, but also increase the risk of mortality among patients who infected by S. aureus. These findings terminated the trial of V710 vaccine.
Currently, the studies on the S. aureus vaccine have focused on some other antigens, such as SE, HLα, TSST 1, and PVL. Toxins of S. aureus were also considered as possible protective antigens. Up to now, no S. aureus vaccine has been proved to be protective, yet. Several clinical trials with novel S. aureus vaccine candidates are still ongoing.
Introduction of the experimental vaccines
The experimental vaccine RSAV in this study is a novel vaccine by Chengdu Olymvax Biopharmaceuticals Inc and Third Military Medical University. RSAV contains five different antigens, more than any other vaccine candidates do. These antigens include IsdB, Hla, SpA, SEB and MntC, which contains bacterial toxin molecules, membrane proteins, and the proteins closely related to the growth and metabolism of S. aureus (1,11-13). Thus, vaccine contains these antigens is supposed to be able to elicit various antibodies to inhibit or block multiple points of bacterial pathogenesis. These recombinant proteins are genetically modified in order to get rid of toxicity but maintain their immunogenicity. RSAV showed a good protection of 88.3% in mice challenging models in pre-clinical studies, which was much higher comparing with a 58% protection reported in previous studies of V710.
The priority target population of S. aureus vaccine should be the aged population, immunosuppressed patients or high risk population with some underling conditions, especially those with open injury or undergoing an operation, ICU patients, long-term hemodialysis patients, large area burn patients, and so on. Clinical epidemiological data showed that the high-risk period of S. aureus infection is about 7 days after the injury or surgery (14). Therefore, we expected that an intensive immunization regimen as three-dose at day 0, 3, 7 or four-dose at day 0, 3, 7, 14 could elicit a quick and strong protection.
Benefits and risks
Possible risks for taking part in this study:
- As a new vaccine first applied in human, there are little available data about its safety profile in human. Therefore, the participants will be closely observed for any adverse reactions/events after vaccination. The common adverse reactions after vaccination include injection-site pain, erythema, swelling, induration, itchiness, and some systematic adverse reaction such as fever, diarrhea, or other discomfort. Generally, these adverse reactions are mild and usually relieve or disappear within 3 days without treatment. Besides, ecchymosis and mild pain will probably occur at blood sampling site.
- Participants in this study may obtain the immune responses against S. aureus and be protected from diseases caused by S. aureus infection in a certain period of time. But the vaccination cannot provide a 100% guarantee for the protection against S. aureus. Participants in this study will get a reasonable compensation for the time, energy, extra expenses (including travel expenses), and any potential risk for participating in this study.
Disease background
S. aureus, a gram positive bacteria, is one of a major cause of hospital-associated and community-associated infections worldwide. These infections caused by S. aureus ranged from minor skin and soft tissue infections to the major life-threatening invasive infections (1). The frequencies of both community-acquired and hospital-acquired S. aureus infections have increased steadily in recent years. Treatment of these infections caused by S. aureus has become more and more difficult because of the emergence of multidrug-resistant strains (4).
A comprehensive survey conducted in clinical sites in the USA, Canada, Europe, found that S. aureus accounted for 22% of all blood infections (8,929 of 40,497 infections), 23.2% of all lower respiratory tract (3,371/14,552 infections) and 39.2% (2,928/7,474 infections) of all skin and soft tissue infections (2). In China, S. aureus was the most common clinical gram-positive bacteria with the highest isolated rate, and the methicillin-resistant S. aureus (MRSA) accounted for 44.6% of all the isolated strains (3). It is critical to develop an effective vaccine to control the infection caused by S. aureus and reduce the usage of antibiotics in order to decrease the drug resistance of S. aureus.
Currently, there is no effective vaccine against S. aureus has been reported. Early clinical trials of several vaccine candidates such as Nabi’s StaphVax and PentaStaph, Intercell’s/Merck’s V710, VRi’s SA75 were all found to be invalid against infectious disease caused by S. aureus (7). Despite the fact that some vaccine candidates were highly immunogenic and able to elicit strong antibody responses, but no significant protection was noted. Moreover, some vaccine candidates even aggravated the risk of infection associated with S. aureus after vaccination (7). Up to now, none of these candidates was effective in preventing S. aureus infection.
The novel experimental vaccine under evaluation in this study, contains five antigens: the alpha hemolysin (Hla), iron-regulated surface determinant B (IsdB), S. aureus protein A (SpA), enterotoxin B (SEB), manganese transport protein C (MntC). In preclinical studies of this vaccine candidate, a total of 16,000 mice and 40 cynomolgus monkeys were tested. The vaccine was safe and immunogenic, showing a protection of over 85%. This is a phase 1 trial to evaluate the safety and tolerability and preliminary immunogenicity (including humoral immunity and cellular immunity) of this novel RSAV in healthy adults aged between 18–65 years.
Pathogeny characteristics
S. aureus could be typed through different methods including serological typing, phage typing, drug-resistant spectrum typing, genetic typing etc. The typing of S. aureus can be used to study the pathogenicity, drug resistance of S. aureus, and the relationship between the epidemiological characteristics and bacterial identification. According to the differences of enterotoxin antigenicity and isoelectric point, the S. aureus can be typed into A, B, C1, C2, C3, D, E, and F. Of them, S. aureus type A and B are most common. This bacteria has multiple antigens or cross-reactive antigens, which are poor in immunogenicity.
S. aureus as pathogenic bacteria widely exist in the environment. It can produce a variety of pathogenic toxin associated with various infectious diseases (4). Human are the main reservoir of S. aureus, and the bacteria is frequently colonized in the nose, respiratory tract, skin wounds, trachea incision site and even on the normal skin. The carriers of S. aureus have no symptom of infections, but are the important source of infection, causing nosocomial infection or community infection through nasal pharyngeal airway, skin wound, blood, or other routines. In addition, the S. aureus also can cause infection through contaminated food.
At hospital, population with the most high-risk of S. aureus infection are the surgery patients, ICU patients, long-term hemodialysis patients, large area burn patients, diabetes and organization transplantation patients. In community, children, adolescents, soldiers, and athletes have an injury of skin soft tissue and immunocompromised elderly people are also high risk group for the community-source S. aureus infection.
Vaccine development in other countries
Up to now, clinical trials of several S. aureus vaccines have been launched by biopharmaceutical companies, including Merck, Pfizer, Novartis, GlaxoSmithKline (GSK), The National Institute of Allergy and Infectious Diseases (NIAID) and so on (Table S1) (10).
Nabi Company (the U.S.) developed a novel capsular polysaccharide S. aureus vaccine StaphVax. The phase 3 clinical trials of StaphVax involved in 3,600 patients with hemodialysis and followed for S. aureus infection after vaccination from 3 to 35 weeks. Results showed that the StaphVax could not provide protection against S. aureus infection (5). In another phase 2 clinical trial for passive immunization of CP5 and CP8 antibodies which was isolated from healthy volunteers vaccinated with StaphVax (StaphVax indued a high level of CP5 and CP8 antibodies in serum), confirmed that CP5 and CP8 antibodies were not able to reduce the risk of S. aureus bacteremia (6). The results indicated that vaccine only contained S. aureus capsular polysaccharide antigen may not be able to provide protection against S. aureus infection.
Merck developed a recombinant subunit vaccine V710, which contains IsdB as antigen. IsdB is a conservative protein wildly existed in different isolates of MRSA, which is an anchor protein of cell wall and plays a key role in absorption of heme iron. In animal models, recombinant mice immunized with IsdB demonstrated a higher survival rate when exposed to lethal challenge with S. aureus. Besides, IsdB also showed a good immunogenicity in rhesus monkey. Moreover, a monoclonal antibody CS-D7 against IsdB showed a significant protection in mice challenging models. These results indicated that IsdB was a promising antigen for S. aureus vaccine. Based on above evidences, Merck cooperated with Intercell to develop the vaccine candidate V710 using IsdB as antigen. Between December 2007 and August 2011, a double-blind, randomized, event-driven trial in heart surgery patients was conducted at 165 sites in 26 countries. However, an interim analysis results showed that no significant difference between the vaccine and placebo recipients in terms of S. aureus infection incidence, even though V710 induced a strong immune response in the vaccine recipients (9). V710 could not only be able to reduce the rate of serious postoperative S. aureus infections among the patients, but also increase the risk of mortality among patients who infected by S. aureus. These findings terminated the trial of V710 vaccine.
Currently, the studies on the S. aureus vaccine have focused on some other antigens, such as SE, HLα, TSST 1, and PVL. Toxins of S. aureus were also considered as possible protective antigens. Up to now, no S. aureus vaccine has been proved to be protective, yet. Several clinical trials with novel S. aureus vaccine candidates are still ongoing.
Introduction of the experimental vaccines
The experimental vaccine RSAV in this study is a novel vaccine by Chengdu Olymvax Biopharmaceuticals Inc and Third Military Medical University. RSAV contains five different antigens, more than any other vaccine candidates do. These antigens include IsdB, Hla, SpA, SEB and MntC, which contains bacterial toxin molecules, membrane proteins, and the proteins closely related to the growth and metabolism of S. aureus (1,11-13). Thus, vaccine contains these antigens is supposed to be able to elicit various antibodies to inhibit or block multiple points of bacterial pathogenesis. These recombinant proteins are genetically modified in order to get rid of toxicity but maintain their immunogenicity. RSAV showed a good protection of 88.3% in mice challenging models in pre-clinical studies, which was much higher comparing with a 58% protection reported in previous studies of V710.
The priority target population of S. aureus vaccine should be the aged population, immunosuppressed patients or high risk population with some underling conditions, especially those with open injury or undergoing an operation, ICU patients, long-term hemodialysis patients, large area burn patients, and so on. Clinical epidemiological data showed that the high-risk period of S. aureus infection is about 7 days after the injury or surgery (14). Therefore, we expected that an intensive immunization regimen as three-dose at day 0, 3, 7 or four-dose at day 0, 3, 7, 14 could elicit a quick and strong protection.
Benefits and risks
Possible risks for taking part in this study:
- As a new vaccine first applied in human, there are little available data about its safety profile in human. Therefore, the participants will be closely observed for any adverse reactions/events after vaccination. The common adverse reactions after vaccination include injection-site pain, erythema, swelling, induration, itchiness, and some systematic adverse reaction such as fever, diarrhea, or other discomfort. Generally, these adverse reactions are mild and usually relieve or disappear within 3 days without treatment. Besides, ecchymosis and mild pain will probably occur at blood sampling site.
- Participants in this study may obtain the immune responses against S. aureus and be protected from diseases caused by S. aureus infection in a certain period of time. But the vaccination cannot provide a 100% guarantee for the protection against S. aureus. Participants in this study will get a reasonable compensation for the time, energy, extra expenses (including travel expenses), and any potential risk for participating in this study.
Study objectives
Primary objective
To evaluate the safety and tolerability of the RSAV in healthy adults aged 18–65 years in China.
Secondary objective
To evaluate the primarily immunogenicity of the RSAV in healthy adults aged 18–65 years in China.
Primary objective
To evaluate the safety and tolerability of the RSAV in healthy adults aged 18–65 years in China.
Secondary objective
To evaluate the primarily immunogenicity of the RSAV in healthy adults aged 18–65 years in China.
Study design
Study design and method description
This is a single-center, randomized, double-blind, placebo-controlled clinical trial. A two-stage randomization will be applied in this study. In the first stage randomization, a total of 144 participants will be randomly assigned to the low-dose group, middle- dose group, high-dose group, and placebo group in a ratio of 1:1:1:1, with 36 participants in each group. The second stage randomization is a nested randomization for the fourth dose within each original treatment group: 36 participants in each treatment group will be randomly assigned to receive corresponding vaccine dose or placebo in a ratio of 2:1.
This is a double-blinded study. Both participants and investigators will be masked to the treatment allocation. Sponsor’s personnel who directly involved in the conduct of this study (e.g., site monitors, medical monitors), laboratory personnel, Data and Safety Monitoring Board (DSMB) will also be blinded to the participant’s treatment allocations.
The investigational vaccines at different dosage and the placebo will be randomized and coded by an independent statistician from the School of Public Health, Southeast University, People’s Republic of China, using SAS 9.3 software. All the randomized vaccine and placebo doses will be identical in the package, and labeled with randomized numbers which are the only identifier for each dose. Therefore, all the vaccine and placebo doses are blindly coded. The personnel who participate in blinding shall not take a part in any other process of the clinical trial, also shall not disclose the contents of blinding to any personnel to participate in clinical trial work.
The serological data, which would lead to the unblinding of the treatment groups, will not be available during the course of the study to any investigator or any person involved in the clinical conduct of the study. The independent statistician should produce a copy of emergency blinding code which should note the title of program and the character “To Be Opened Only in Case of Emergency”.
During the study, the breaking of the treatment code is forbidden, except in the event of a medical emergency or unexpected serious adverse event associated with the use of the investigational product, when the investigator believes it is necessary to determine the treatment code in order to initiate appropriate treatment. If knowledge of the treatment code is required, the investigator will open only the specific participant’s emergency blinding code. A signature, date, time and reason will be written on the opened code envelope and the participant with this code has to be withdrawn from this study.
The investigator will assess the relationship between the adverse event and the investigational products before the treatment code is unblinded. The investigator will immediately notify the sponsors at the 24 hours emergency call number (Hao Zeng +86-13983488826) when the treatment code is broken on any participant for any reason during the study. The reason for the treatment code being broken must be also documented in the participant’s medical records and in the case report forms (CRF).
At the end of the study, all code envelopes (intact and opened) must be accounted for and are to be collected by the monitor to be destroyed.
Study endpoints
Safety: occurrence of solicited adverse reactions within 21 days after vaccination.
- Safety:
- Abnormal change of safety laboratory measures (routine blood test and blood biochemical test) on day 3, day 7, day 10, day 14, and day 17 compared with the baseline;
- Occurrence of unsolicited adverse reactions within 42 days after the first vaccination;
- Occurrence of serious adverse events (SAEs) during the whole follow-up period (6 months).
- Immunogenicity:
- Antibody geometric mean titers (GMTs) of antigen-specific antibodies in serum at day 7, day 14, day 21, day 42, month 3, and month 6 post-vaccination;
- The geometric mean fold increase (GMFI) of antigen-specific antibodies in serum at day 7, day 14, day 21, day 42, month 3, and month 6 post-vaccination;
- Seroconversion rate of antigen-specific antibodies in serum t day 7, day 14, day 21, day 42, month 3, and month 6 post-vaccination.
- S. aureus colonization:
- The nasal S. aureus colonization in participant’s nose at day 3, day 7, day 10, day 7, day 14, day 17, day 21, day 42, month 3, and month 6 post-vaccination.
- Functional antibody measurement:
- Functional antibody titres at day 7, day 14, day 21, day 42, month 3, and month 6 post-vaccination.
- Cellular immune response:
- Cellular immune responses at day 7, day 14, day 21, day 42, month 3, month 6 post-vaccination.
Study procedures
Participants will attend a total of 11 visits, including the screening visit (V0) before the vaccination and 10 site visits during a study period of 6 months (Tables S2-S4).
Sample size
According to the China guidelines for vaccine clinical trial, the sample size of a phase 1 clinical trial should be around 20–30. We did not calculated the sample size of this clinical trials specifically according to statistical power. We are going to recruit 36 participants for each treatment group. A total of 144 participants will be recruited.
Criteria for pausing the study or an early termination
On the first two days of enrollment, no more than 12 participants will be recruited and vaccinated each day. Then no more than 24 participants will be recruited and vaccinated in each day in the following recruiting period.
DSMB will review the reported safety data during the study period. If an increase of risk for participants is noticed, the DSMB should promptly inform the principle investigator and sponsors. Sponsors, investigators and DSMB will have a panel meeting, and then DSMB will make final decision whether need to pause or call an early termination of the study.
Administration of study injections and new enrollments will be paused, if:
- One serious adverse event may be associated with vaccination, or
- Occurrence of grade 3 adverse reaction may be associated with vaccination in 10% of participants or more (including injection-site reaction, systemic reaction, and change of the safety laboratory measures).
The study may come to an early termination, if:
- One vaccination-associated serious adverse event, or
- Occurrence of grade 3 adverse events associated with vaccination in 15% of participants or more (including injection-site reaction, systemic reaction, and change of the safety laboratory measures), or
- Required by sponsor, or
- Required by regulatory authority, or
- Required by institutional review board (IRB).
Duration of study
The whole follow-up period for each participant will be 6 months.
Study design and method description
This is a single-center, randomized, double-blind, placebo-controlled clinical trial. A two-stage randomization will be applied in this study. In the first stage randomization, a total of 144 participants will be randomly assigned to the low-dose group, middle- dose group, high-dose group, and placebo group in a ratio of 1:1:1:1, with 36 participants in each group. The second stage randomization is a nested randomization for the fourth dose within each original treatment group: 36 participants in each treatment group will be randomly assigned to receive corresponding vaccine dose or placebo in a ratio of 2:1.
This is a double-blinded study. Both participants and investigators will be masked to the treatment allocation. Sponsor’s personnel who directly involved in the conduct of this study (e.g., site monitors, medical monitors), laboratory personnel, Data and Safety Monitoring Board (DSMB) will also be blinded to the participant’s treatment allocations.
The investigational vaccines at different dosage and the placebo will be randomized and coded by an independent statistician from the School of Public Health, Southeast University, People’s Republic of China, using SAS 9.3 software. All the randomized vaccine and placebo doses will be identical in the package, and labeled with randomized numbers which are the only identifier for each dose. Therefore, all the vaccine and placebo doses are blindly coded. The personnel who participate in blinding shall not take a part in any other process of the clinical trial, also shall not disclose the contents of blinding to any personnel to participate in clinical trial work.
The serological data, which would lead to the unblinding of the treatment groups, will not be available during the course of the study to any investigator or any person involved in the clinical conduct of the study. The independent statistician should produce a copy of emergency blinding code which should note the title of program and the character “To Be Opened Only in Case of Emergency”.
During the study, the breaking of the treatment code is forbidden, except in the event of a medical emergency or unexpected serious adverse event associated with the use of the investigational product, when the investigator believes it is necessary to determine the treatment code in order to initiate appropriate treatment. If knowledge of the treatment code is required, the investigator will open only the specific participant’s emergency blinding code. A signature, date, time and reason will be written on the opened code envelope and the participant with this code has to be withdrawn from this study.
The investigator will assess the relationship between the adverse event and the investigational products before the treatment code is unblinded. The investigator will immediately notify the sponsors at the 24 hours emergency call number (Hao Zeng +86-13983488826) when the treatment code is broken on any participant for any reason during the study. The reason for the treatment code being broken must be also documented in the participant’s medical records and in the case report forms (CRF).
At the end of the study, all code envelopes (intact and opened) must be accounted for and are to be collected by the monitor to be destroyed.
Study endpoints
Safety: occurrence of solicited adverse reactions within 21 days after vaccination.
- Safety:
- Abnormal change of safety laboratory measures (routine blood test and blood biochemical test) on day 3, day 7, day 10, day 14, and day 17 compared with the baseline;
- Occurrence of unsolicited adverse reactions within 42 days after the first vaccination;
- Occurrence of serious adverse events (SAEs) during the whole follow-up period (6 months).
- Immunogenicity:
- Antibody geometric mean titers (GMTs) of antigen-specific antibodies in serum at day 7, day 14, day 21, day 42, month 3, and month 6 post-vaccination;
- The geometric mean fold increase (GMFI) of antigen-specific antibodies in serum at day 7, day 14, day 21, day 42, month 3, and month 6 post-vaccination;
- Seroconversion rate of antigen-specific antibodies in serum t day 7, day 14, day 21, day 42, month 3, and month 6 post-vaccination.
- S. aureus colonization:
- The nasal S. aureus colonization in participant’s nose at day 3, day 7, day 10, day 7, day 14, day 17, day 21, day 42, month 3, and month 6 post-vaccination.
- Functional antibody measurement:
- Functional antibody titres at day 7, day 14, day 21, day 42, month 3, and month 6 post-vaccination.
- Cellular immune response:
- Cellular immune responses at day 7, day 14, day 21, day 42, month 3, month 6 post-vaccination.
Study procedures
Participants will attend a total of 11 visits, including the screening visit (V0) before the vaccination and 10 site visits during a study period of 6 months (Tables S2-S4).
Sample size
According to the China guidelines for vaccine clinical trial, the sample size of a phase 1 clinical trial should be around 20–30. We did not calculated the sample size of this clinical trials specifically according to statistical power. We are going to recruit 36 participants for each treatment group. A total of 144 participants will be recruited.
Criteria for pausing the study or an early termination
On the first two days of enrollment, no more than 12 participants will be recruited and vaccinated each day. Then no more than 24 participants will be recruited and vaccinated in each day in the following recruiting period.
DSMB will review the reported safety data during the study period. If an increase of risk for participants is noticed, the DSMB should promptly inform the principle investigator and sponsors. Sponsors, investigators and DSMB will have a panel meeting, and then DSMB will make final decision whether need to pause or call an early termination of the study.
Administration of study injections and new enrollments will be paused, if:
- One serious adverse event may be associated with vaccination, or
- Occurrence of grade 3 adverse reaction may be associated with vaccination in 10% of participants or more (including injection-site reaction, systemic reaction, and change of the safety laboratory measures).
The study may come to an early termination, if:
- One vaccination-associated serious adverse event, or
- Occurrence of grade 3 adverse events associated with vaccination in 15% of participants or more (including injection-site reaction, systemic reaction, and change of the safety laboratory measures), or
- Required by sponsor, or
- Required by regulatory authority, or
- Required by institutional review board (IRB).
Duration of study
The whole follow-up period for each participant will be 6 months.
Participants
Participants selection
The following inclusion and exclusion criteria will be used to select the eligible participants for this study.
Inclusion criteria
- Healthy volunteers aged between 18 and 65 years.
- Willing to comply with the clinical trial protocol and complete all the visits planed during the study period.
- Able to understand the content of informed consent and willing to sign the informed consent.
- Able to complete the diary card independently.
- For females only (18–49 years), a negative urine pregnancy test and willing to practice continuous effective contraception during the study.
- Axillary temperature ≤37.0 °C.
Exclusion criteria
First immunization exclusion standard:
- Prior receipt of S. aureus vaccine;
- Any confirmed S. aureus infection disease in the past 12 month;
- History of asthma, hereditary angioneurotic edema, diabetes, malignancy or other serious disease. Participation in the clinical trial is likely to increase the disease risk and interfere with the observation of clinical trial index;
- Prior blood donation or blood loss over 400 mL in the last 3 months;
- Coagulation disorders (coagulation factor deficiency, coagulopathy or platelet disorder) diagnosed by doctors, or obvious bruises or blood coagulation noticed;
- History of allergic disease likely to be exacerbated by any component of the vaccine, including allergy, urticaria, respiratory difficulty, angioneurotic edema or abdominal pain;
- Any autoimmune disease or immunodeficient state, parents, brother and sister with autoimmune disease or immunodeficient disease.
- Taking immunoglobulins and/or any blood products within the last 12 months;
- Asplenia, functional asplenia or asplenia caused by any situation or splenectomy;
- Any acute disease or acute attack of chronic disease in last 7 days;
- History of thyroidectomy or thyroid disease requiring treatment in the last 12 months;
- Immunosuppressor, cytotoxic therapy, inhaled corticosteroid (excluding corticosteroids spray treatment of allergic rhinitis, acute and non-concurrent corticosteroids treatment);
- Participation in another research study involving receipt of an investigational product in the last 30 days;
- Woman who is breast-feeding;
- Prior administration of attenuated vaccine in last 28 days;
- Prior administration of subunit vaccine, inactivated vaccine or allergic therapy in last 14 days;
- Current anti-tuberculosis prophylaxis or therapy;
- Any other conditions may compromise the safety or availability of participants in the judgment of the investigator.
Withdraw from the study
If any of the following condition occur in the participants during the study, investigator can decide whether the participants need to withdraw from the study:
- Severe violation of the protocol;
- Have some food and/or medicine can interfere with the immune response during the observation period of the primary objectives (day 0 to day 28);
- Unwilling to continue the study and request to withdraw;
- Any intolerable adverse events (related to vaccination or not);
- New diagnosed disorder which makes the participants not suitable to continue the study;
- The presence of any condition needs to be withdrawn determined by investigators.
Participants selection
The following inclusion and exclusion criteria will be used to select the eligible participants for this study.
Inclusion criteria
- Healthy volunteers aged between 18 and 65 years.
- Willing to comply with the clinical trial protocol and complete all the visits planed during the study period.
- Able to understand the content of informed consent and willing to sign the informed consent.
- Able to complete the diary card independently.
- For females only (18–49 years), a negative urine pregnancy test and willing to practice continuous effective contraception during the study.
- Axillary temperature ≤37.0 °C.
Exclusion criteria
First immunization exclusion standard:
- Prior receipt of S. aureus vaccine;
- Any confirmed S. aureus infection disease in the past 12 month;
- History of asthma, hereditary angioneurotic edema, diabetes, malignancy or other serious disease. Participation in the clinical trial is likely to increase the disease risk and interfere with the observation of clinical trial index;
- Prior blood donation or blood loss over 400 mL in the last 3 months;
- Coagulation disorders (coagulation factor deficiency, coagulopathy or platelet disorder) diagnosed by doctors, or obvious bruises or blood coagulation noticed;
- History of allergic disease likely to be exacerbated by any component of the vaccine, including allergy, urticaria, respiratory difficulty, angioneurotic edema or abdominal pain;
- Any autoimmune disease or immunodeficient state, parents, brother and sister with autoimmune disease or immunodeficient disease.
- Taking immunoglobulins and/or any blood products within the last 12 months;
- Asplenia, functional asplenia or asplenia caused by any situation or splenectomy;
- Any acute disease or acute attack of chronic disease in last 7 days;
- History of thyroidectomy or thyroid disease requiring treatment in the last 12 months;
- Immunosuppressor, cytotoxic therapy, inhaled corticosteroid (excluding corticosteroids spray treatment of allergic rhinitis, acute and non-concurrent corticosteroids treatment);
- Participation in another research study involving receipt of an investigational product in the last 30 days;
- Woman who is breast-feeding;
- Prior administration of attenuated vaccine in last 28 days;
- Prior administration of subunit vaccine, inactivated vaccine or allergic therapy in last 14 days;
- Current anti-tuberculosis prophylaxis or therapy;
- Any other conditions may compromise the safety or availability of participants in the judgment of the investigator.
Withdraw from the study
If any of the following condition occur in the participants during the study, investigator can decide whether the participants need to withdraw from the study:
- Severe violation of the protocol;
- Have some food and/or medicine can interfere with the immune response during the observation period of the primary objectives (day 0 to day 28);
- Unwilling to continue the study and request to withdraw;
- Any intolerable adverse events (related to vaccination or not);
- New diagnosed disorder which makes the participants not suitable to continue the study;
- The presence of any condition needs to be withdrawn determined by investigators.
Investigational product
Investigational vaccine
Experimental vaccine is RSAV developed by Chengdu Olymvax Biopharmaceuticals Inc. and Third Military Medical University. Vaccine contains HI, SpA5, mSEB, MntC recombinant proteins and aluminum phosphate adjuvants. HI is a fusion protein consisted of Hla and IsdB. The final product is 0.6 mL mixed suspension per vial for injection.
Low dose experimental vaccine: RSAV (15 µg/single-protein), 0.6 mL/vial, 15 µg HI, SpA5, mSEB, and MntC protein, 1.952 mg aluminum phosphate (0.432 mg aluminum), batch: 20150702, valid date: 28 July 2017.
Medium dose experimental vaccine: RSAV (30 µg/single-protein), 0.6 mL/vial, 30 µg HI, SpA5, mSEB, and MntC protein, 1.952 mg aluminum phosphate (0.432 mg aluminum), batch: 20150703, valid date: 28 July 2017.
High dose experimental vaccine: RSAV (60 µg/single-protein), 0.6 mL/vial, 60 µg HI, SpA5, mSEB, and MntC protein, 1.952 mg aluminum phosphate (0.432 mg aluminum), batch: 20150704, valid date: 28 July 2017.
Placebo: excipients, protein-free, 1.952 mg aluminum phosphate (0.432 mg aluminum), batch: 20150704, valid date: 28 July 2017.
Administration
The injection will be given intramuscularly in the deltoid muscle in participants. Before injection, 75% alcohol is used for disinfection at the injection site, intramuscular vaccination will be administrated several minutes later. Shaking the vaccine before use. No intravascular, intradermal or subcutaneous injection is allowed with the investigational vaccine. At the clinic site, during the vaccination and the 30-minute safety observation after vaccination, appropriate emergency medical equipment and doctors should put on standby in case acute allergic reaction after injection or other emergency condition. Only the investigator or medically qualified designee will give the injection.
Transportation and storage
The investigational vaccines should be stored in a safe and locked refrigerator where nobody can get them without authorization. Sponsors should evaluate the storage conditions of the research center before the study to make sure that the vaccines will be stored under appropriate condition during the study.
The vaccine storage temperature should be kept +2 to +8 °C during the transportation from the sponsors to the clinical research center. The temperature should be checked during transportation by suitable temperature monitor in instrument. When receiving the vaccines at the study center, the receiver should check the vaccines’ number, quality and the maintenance of cold chain during transportation, and fill in the vaccine receipt form.
The temperature should be recorded every day during storage by temperature monitoring instrument and documented each workday manually (once in the morning, once in the afternoon). Frozen indicating instrument should be put near the vaccines to monitor if they are frozen.
Investigational vaccine
Experimental vaccine is RSAV developed by Chengdu Olymvax Biopharmaceuticals Inc. and Third Military Medical University. Vaccine contains HI, SpA5, mSEB, MntC recombinant proteins and aluminum phosphate adjuvants. HI is a fusion protein consisted of Hla and IsdB. The final product is 0.6 mL mixed suspension per vial for injection.
Low dose experimental vaccine: RSAV (15 µg/single-protein), 0.6 mL/vial, 15 µg HI, SpA5, mSEB, and MntC protein, 1.952 mg aluminum phosphate (0.432 mg aluminum), batch: 20150702, valid date: 28 July 2017.
Medium dose experimental vaccine: RSAV (30 µg/single-protein), 0.6 mL/vial, 30 µg HI, SpA5, mSEB, and MntC protein, 1.952 mg aluminum phosphate (0.432 mg aluminum), batch: 20150703, valid date: 28 July 2017.
High dose experimental vaccine: RSAV (60 µg/single-protein), 0.6 mL/vial, 60 µg HI, SpA5, mSEB, and MntC protein, 1.952 mg aluminum phosphate (0.432 mg aluminum), batch: 20150704, valid date: 28 July 2017.
Placebo: excipients, protein-free, 1.952 mg aluminum phosphate (0.432 mg aluminum), batch: 20150704, valid date: 28 July 2017.
Administration
The injection will be given intramuscularly in the deltoid muscle in participants. Before injection, 75% alcohol is used for disinfection at the injection site, intramuscular vaccination will be administrated several minutes later. Shaking the vaccine before use. No intravascular, intradermal or subcutaneous injection is allowed with the investigational vaccine. At the clinic site, during the vaccination and the 30-minute safety observation after vaccination, appropriate emergency medical equipment and doctors should put on standby in case acute allergic reaction after injection or other emergency condition. Only the investigator or medically qualified designee will give the injection.
Transportation and storage
The investigational vaccines should be stored in a safe and locked refrigerator where nobody can get them without authorization. Sponsors should evaluate the storage conditions of the research center before the study to make sure that the vaccines will be stored under appropriate condition during the study.
The vaccine storage temperature should be kept +2 to +8 °C during the transportation from the sponsors to the clinical research center. The temperature should be checked during transportation by suitable temperature monitor in instrument. When receiving the vaccines at the study center, the receiver should check the vaccines’ number, quality and the maintenance of cold chain during transportation, and fill in the vaccine receipt form.
The temperature should be recorded every day during storage by temperature monitoring instrument and documented each workday manually (once in the morning, once in the afternoon). Frozen indicating instrument should be put near the vaccines to monitor if they are frozen.
Safety assessments and adverse event reporting
Adverse event and adverse reaction
An adverse event (AE) is any untoward medical occurrence in a participant administered an investigational product and which does not necessarily have a causal relationship with this treatment. An adverse reaction (AR) is all untoward and unintended responses to a medical product related to any dose administered.
An unexpected adverse reaction is an adverse reaction, the nature or severity of which is not consistent with the applicable product information:
- Investigator’s brochure for an unauthorized experimental product;
- Summary of product characteristics for an authorized product;
- For safety assessments, the primary outcome measure is the solicited adverse reactions within 21 days;
- Participants will remain in the clinic to observe for the occurrence of any adverse events for at least 30 minutes after receipt of vaccination. For the next 42 days following vaccination, any injection-site adverse events and systematic adverse events will be recorded by the participants on the diary cards. Blood samples collected from participants before the vaccination, on day 3, 7, 10, 14, 17, after vaccination will be tested for any significant changes of laboratory testing from baseline. The serious adverse events during the whole study period;
- All adverse events (including laboratory measures) will be graded according to “The standard guidelines for adverse reactions grading of vaccine clinical trials” issued by China state Food and Drug Administration (SFDA). The presence of solicited and unsolicited adverse events and any serious adverse events will be described in terms of the incidence, intensity and relation to vaccination. The incidence of adverse events will be based on the most severe response, and expressed in terms of the number and proportion of individuals who had adverse events in each group;
Grading for adverse events
Pain (at the injection site):
- Grade 1 (mild) = mild pain when the injection site was touched, does not interfere with activities;
- Grade 2 (moderate) = moderate pain, taking non-narcotic pain killer;
- Grade 3 (severe) = significant; prevents daily activity, taking narcotic pain killer;
- Grade 4 (potentially life threatening) = emergency or hospitalization.
Mucocutaneous (at the injection site):
- Grade 1 (mild) = redness and itch;
- Grade 2 (moderate) = scattered rash, rash-like tetter, dry, desquamation;
- Grade 3 (severe) = blister-like eruptions, moist, desquamation or ulceration;
- Grade 4 (potentially life threatening) = peeling dermatitis, or erythema multiforme, or like Stevens-Johnsons syndrome.
Induration (at the injection site):
- Grade 1 (mild) =2.5–5 cm and does not interfere with activity;
- Grade 2 (moderate) =5.1–10 cm or interferes with activity;
- Grade 3 (severe) = >10 cm or prevents daily activity;
- Grade 4 (potentially life threatening) = necrosis.
Redness (at the injection site):
- Grade 1 (mild) = 2.5–5 cm;
- Grade 2 (moderate) =5.1–10 cm;
- Grade 3 (severe) = >10 cm;
- Grade 4 (potentially life threatening) = necrosis or exfoliative dermatitis.
Swelling (at the injection site):
- Grade 1 (mild) = 2.5–5 cm and does not interfere with activity;
- Grade 2 (moderate) = 5.1–10 cm or interferes with activity;
- Grade 3 (severe) = >10 cm or prevents daily activity;
- Grade 4 (potentially life threatening) = necrosis.
Itch (at the injection site):
- Grade 1 (mild) = slight itch at the injection site;
- Grade 2 (moderate) = moderate itch on the injection arm;
- Grade 3 (severe) = significant systemic itch.
Fever (axillary temperature):
- Grade 1 (mild) = ≥ 37.1 to ≤ 37.5 °C;
- Grade 2 (moderate) = ≥ 37.6 to ≤ 39.0 °C;
- Grade 3 (severe) = >39.0 °C.
Allergic reactions:
- Grade 1 (mild) = pruritus without rashes;
- Grade 2 (moderate) = localized urticarial;
- Grade 3 (severe) = general urticaria, angioedema;
- Grade 4 (potentially life threatening) = severe allergic reactions, emergency or hospitalization.
Headache:
- Grade 1 (mild) = mild symptoms that do not interfere with normal daily activities, need no treatment;
- Grade 2 (moderate) = transient headache, symptoms that have a mild impact on normal daily activities, may need some pain killer;
- Grade 3 (severe) = symptoms that have a significant impact on normal daily activities, and need treatment;
- Grade 4 (potentially life threatening) = stubborn headache stubborn headache, need hospitalization.
Fatigue:
- Grade 1 (mild) =mild symptoms that do not interfere with normal daily activities, last ≤48 hours;
- Grade 2 (moderate) = symptoms that have an impact on normal daily activities, last >48 hours;
- Grade 3 (severe) = symptoms that have an impact on normal daily activities, last >72 hours;
- Grade 4 (potentially life threatening) =emergency or hospitalization.
Vomiting:
- Grade 1 (mild) = once to twice times per 24 hours, do not interfere with normal food intake and no impairment of activities;
- Grade 2 (moderate) = twice to five times per 24 hours, food intake significantly reduced or limitations of activities;
- Grade 3 (severe) = > six times per 24 hours, little food intake, necessity of intravenous infusion;
- Grade 4 (potentially life threatening) = >could not eat or drink for more than 24 hours.
Diarrhea:
- Grade 1 (mild) = slightly or transient, twice to three times of watery stools per day, or continuously slight diarrhea within one week;
- Grade 2 (moderate) = moderate or continuously, four to five times per day or diarrhea, symptom continus more than one week;
- Grade 3 (severe) = > six times of watery stool per day, or bloody stool, postural hypotension, electrolyte imbalance and necessity of intravenous infusion >2 liter;
- Grade 4 (potentially life threatening) = hospitalization due to hypertensive shock.
Myalgia:
- Grade 1 (mild) = transient, no interference with activity;
- Grade 2 (moderate) = some interference with daily activity;
- Grade 3 (severe) = significant; prevents daily activity, need treatment;
- Grade 4 (potentially life threatening) = muscular necrosis, emergency of hospitalization.
Cough:
- Grade 1 (mild) = transient, treatment unnecessary;
- Grade 2 (moderate) = continuous coughs, response to treatment;
- Grade 3 (severe) = paroxysmal coughs, treatment uncontrolled;
- Grade 4 (potentially life threatening) = emergency of hospitalization.
Routine blood and blood biochemical tests will be undertaken by a local hospital (second-class or above), with qualification. Test must be performed using standardized testing instrument and qualified detection kits. Table S5 below shows that indicators of classifying laboratory abnormalities.
Serious adverse event/reaction (SAE)
A serious adverse event/reaction is occurrence of any untoward medical during the whole study period that:
- Results in death;
- Is life-threatening (an event in which the participant is at risk of death at the time of the event; it does not refer to an event which hypothetically might have caused death if it was more severe);
- Results in persistent or significant disability/incapacity;
- Requires hospitalization or prolongation of an existing hospitalization;
- Is a congenital anomaly/birth defect.
In addition, medical and scientific judgment will be exercised in deciding whether other conditions will also be considered serious, such as important medical events that may not be immediately life-threatening or result in death or hospitalization but may jeopardize the participant’s safety or may require intervention to prevent one of the other outcomes listed in the definition above. These will also be considered serious. Examples of such events are intensive treatment in an emergency room or at home for allergic bronchospasm; blood dyscrasias or convulsions that do not result in hospitalization; or development of drug dependency or drug abuse.
Any serious adverse event, including death due to any cause, which occurs during this study, whether or not related to the investigational products, must be reported immediately (within 24 hours of the investigator’s knowledge of the event) by telephone or fax to the sponsors, principle investigator, Jiangsu Provincial Center for Disease Control and Prevention (JSCDC) IRB, and the DSMB, at the following number:
- Principal Investigator: Feng-Cai Zhu, Tel: +86-25-83759418, Fax: +86-25-83759409
- Representative of sponsor: Hao Zeng, Tel: +86-13983488826, Fax: +86-23-68752377
- JSCDC IRB: Hui-Yuan Cai, Tel: +86-25-83759406; Fax: +86-25-83759406
- DSMB: Jie-Lai Xia, Tel: 029-84774858; Fax: 029-84774858
The unblinding of single cases by investigators in the course of the clinical trial will only be performed if relevant for the safety of the participant.
In addition to the initial 24-hour report, a completed, separate SAE report is to be sent to the sponsors, principle investigator, JSCDC IRB, and the DSMB via fax or mail within 48 hours of the event as following report, and an end report after the resolve of the SAE. All SAEs will be recorded on the case report form or other source documents.
It is the responsibility of the sponsor to ensure that all information pertaining to suspected unexpected serious adverse reactions (SUSARs) due to an investigational medicine in a clinical trial.
The sponsor is responsible for the prompt notification to all concerned investigators, the JSCDC IRB and competent authority of all relevant safety information previously described.
Fatal or life-threatening SUSARs will be reported to the competent authority and the JSCDC IRB as soon as possible but no later than seven calendar days after the sponsor has first knowledge of the minimum criteria for expedited reporting.
In each case relevant follow-up information will be sought and a report completed as soon as possible. It will be communicated to the competent authority and the JSCDC IRB within an additional eight calendar days.
All other SUSARs and safety issues deserving expedited reporting must be reported to the competent authority and dependent on national provisions to the JSCDC IRB in the concerned countries as soon as possible but no later than 15 calendar days after the sponsor has first knowledge of the minimum criteria for expedited reporting.
Expedited reporting is not usually required for reactions which are serious but expected, or for non-serious adverse reactions whether expected or not.
Safety data monitoring by DSMB
The DSMB will blindly review safety continuously during the trial and will advise on progression of the trial. Table S6 provides the members of the data and safety monitoring board. It will review the safety data from the trial as generated by the investigators, in order to advise whether or not the study integrity remains intact and whether or not there are any safety concerns worth early notice. DSMB will review the reported safety data in the participants for the first 7 days after vaccination. During the study period, if an increase of risk for participants is noticed, the DSMB should promptly inform the principle investigator and sponsors. Sponsors, investigators and DSMB will have a panel meeting, and then DSMB will make final decision to pause or all an early termination of the study.
Administration of study injections and new enrollments will be paused, if:
- One serious adverse event may be associated with vaccination, or
- Occurrence of grade 3 adverse reaction may be associated with vaccination in 10% of participants or more (including injection-site reaction, systemic reaction, and change of the safety laboratory measures).
The study may come to an early termination, if:
- One vaccination-associated serious adverse event, or
- Occurrence of grade 3 adverse events associated with vaccination in 15% of participants or more (including injection-site reaction, systemic reaction, and change of the safety laboratory measures), or
- Required by sponsor, or
- Required by regulatory authority, or
- Required by institutional review board (IRB).
Withdrawal due to adverse events (see also section on participant withdrawal)
Withdrawal due to AE should be distinguished from withdrawal due to other reasons unrelated to safety issues, according to the definition of AE noted earlier and recorded on the appropriate AE CRF page.
When a participant withdraws due to an SAE, the SAE must be reported in accordance with the reporting requirements defined above.
Adverse event and adverse reaction
An adverse event (AE) is any untoward medical occurrence in a participant administered an investigational product and which does not necessarily have a causal relationship with this treatment. An adverse reaction (AR) is all untoward and unintended responses to a medical product related to any dose administered.
An unexpected adverse reaction is an adverse reaction, the nature or severity of which is not consistent with the applicable product information:
- Investigator’s brochure for an unauthorized experimental product;
- Summary of product characteristics for an authorized product;
- For safety assessments, the primary outcome measure is the solicited adverse reactions within 21 days;
- Participants will remain in the clinic to observe for the occurrence of any adverse events for at least 30 minutes after receipt of vaccination. For the next 42 days following vaccination, any injection-site adverse events and systematic adverse events will be recorded by the participants on the diary cards. Blood samples collected from participants before the vaccination, on day 3, 7, 10, 14, 17, after vaccination will be tested for any significant changes of laboratory testing from baseline. The serious adverse events during the whole study period;
- All adverse events (including laboratory measures) will be graded according to “The standard guidelines for adverse reactions grading of vaccine clinical trials” issued by China state Food and Drug Administration (SFDA). The presence of solicited and unsolicited adverse events and any serious adverse events will be described in terms of the incidence, intensity and relation to vaccination. The incidence of adverse events will be based on the most severe response, and expressed in terms of the number and proportion of individuals who had adverse events in each group;
Grading for adverse events
Pain (at the injection site):
- Grade 1 (mild) = mild pain when the injection site was touched, does not interfere with activities;
- Grade 2 (moderate) = moderate pain, taking non-narcotic pain killer;
- Grade 3 (severe) = significant; prevents daily activity, taking narcotic pain killer;
- Grade 4 (potentially life threatening) = emergency or hospitalization.
Mucocutaneous (at the injection site):
- Grade 1 (mild) = redness and itch;
- Grade 2 (moderate) = scattered rash, rash-like tetter, dry, desquamation;
- Grade 3 (severe) = blister-like eruptions, moist, desquamation or ulceration;
- Grade 4 (potentially life threatening) = peeling dermatitis, or erythema multiforme, or like Stevens-Johnsons syndrome.
Induration (at the injection site):
- Grade 1 (mild) =2.5–5 cm and does not interfere with activity;
- Grade 2 (moderate) =5.1–10 cm or interferes with activity;
- Grade 3 (severe) = >10 cm or prevents daily activity;
- Grade 4 (potentially life threatening) = necrosis.
Redness (at the injection site):
- Grade 1 (mild) = 2.5–5 cm;
- Grade 2 (moderate) =5.1–10 cm;
- Grade 3 (severe) = >10 cm;
- Grade 4 (potentially life threatening) = necrosis or exfoliative dermatitis.
Swelling (at the injection site):
- Grade 1 (mild) = 2.5–5 cm and does not interfere with activity;
- Grade 2 (moderate) = 5.1–10 cm or interferes with activity;
- Grade 3 (severe) = >10 cm or prevents daily activity;
- Grade 4 (potentially life threatening) = necrosis.
Itch (at the injection site):
- Grade 1 (mild) = slight itch at the injection site;
- Grade 2 (moderate) = moderate itch on the injection arm;
- Grade 3 (severe) = significant systemic itch.
Fever (axillary temperature):
- Grade 1 (mild) = ≥ 37.1 to ≤ 37.5 °C;
- Grade 2 (moderate) = ≥ 37.6 to ≤ 39.0 °C;
- Grade 3 (severe) = >39.0 °C.
Allergic reactions:
- Grade 1 (mild) = pruritus without rashes;
- Grade 2 (moderate) = localized urticarial;
- Grade 3 (severe) = general urticaria, angioedema;
- Grade 4 (potentially life threatening) = severe allergic reactions, emergency or hospitalization.
Headache:
- Grade 1 (mild) = mild symptoms that do not interfere with normal daily activities, need no treatment;
- Grade 2 (moderate) = transient headache, symptoms that have a mild impact on normal daily activities, may need some pain killer;
- Grade 3 (severe) = symptoms that have a significant impact on normal daily activities, and need treatment;
- Grade 4 (potentially life threatening) = stubborn headache stubborn headache, need hospitalization.
Fatigue:
- Grade 1 (mild) =mild symptoms that do not interfere with normal daily activities, last ≤48 hours;
- Grade 2 (moderate) = symptoms that have an impact on normal daily activities, last >48 hours;
- Grade 3 (severe) = symptoms that have an impact on normal daily activities, last >72 hours;
- Grade 4 (potentially life threatening) =emergency or hospitalization.
Vomiting:
- Grade 1 (mild) = once to twice times per 24 hours, do not interfere with normal food intake and no impairment of activities;
- Grade 2 (moderate) = twice to five times per 24 hours, food intake significantly reduced or limitations of activities;
- Grade 3 (severe) = > six times per 24 hours, little food intake, necessity of intravenous infusion;
- Grade 4 (potentially life threatening) = >could not eat or drink for more than 24 hours.
Diarrhea:
- Grade 1 (mild) = slightly or transient, twice to three times of watery stools per day, or continuously slight diarrhea within one week;
- Grade 2 (moderate) = moderate or continuously, four to five times per day or diarrhea, symptom continus more than one week;
- Grade 3 (severe) = > six times of watery stool per day, or bloody stool, postural hypotension, electrolyte imbalance and necessity of intravenous infusion >2 liter;
- Grade 4 (potentially life threatening) = hospitalization due to hypertensive shock.
Myalgia:
- Grade 1 (mild) = transient, no interference with activity;
- Grade 2 (moderate) = some interference with daily activity;
- Grade 3 (severe) = significant; prevents daily activity, need treatment;
- Grade 4 (potentially life threatening) = muscular necrosis, emergency of hospitalization.
Cough:
- Grade 1 (mild) = transient, treatment unnecessary;
- Grade 2 (moderate) = continuous coughs, response to treatment;
- Grade 3 (severe) = paroxysmal coughs, treatment uncontrolled;
- Grade 4 (potentially life threatening) = emergency of hospitalization.
Routine blood and blood biochemical tests will be undertaken by a local hospital (second-class or above), with qualification. Test must be performed using standardized testing instrument and qualified detection kits. Table S5 below shows that indicators of classifying laboratory abnormalities.
Serious adverse event/reaction (SAE)
A serious adverse event/reaction is occurrence of any untoward medical during the whole study period that:
- Results in death;
- Is life-threatening (an event in which the participant is at risk of death at the time of the event; it does not refer to an event which hypothetically might have caused death if it was more severe);
- Results in persistent or significant disability/incapacity;
- Requires hospitalization or prolongation of an existing hospitalization;
- Is a congenital anomaly/birth defect.
In addition, medical and scientific judgment will be exercised in deciding whether other conditions will also be considered serious, such as important medical events that may not be immediately life-threatening or result in death or hospitalization but may jeopardize the participant’s safety or may require intervention to prevent one of the other outcomes listed in the definition above. These will also be considered serious. Examples of such events are intensive treatment in an emergency room or at home for allergic bronchospasm; blood dyscrasias or convulsions that do not result in hospitalization; or development of drug dependency or drug abuse.
Any serious adverse event, including death due to any cause, which occurs during this study, whether or not related to the investigational products, must be reported immediately (within 24 hours of the investigator’s knowledge of the event) by telephone or fax to the sponsors, principle investigator, Jiangsu Provincial Center for Disease Control and Prevention (JSCDC) IRB, and the DSMB, at the following number:
- Principal Investigator: Feng-Cai Zhu, Tel: +86-25-83759418, Fax: +86-25-83759409
- Representative of sponsor: Hao Zeng, Tel: +86-13983488826, Fax: +86-23-68752377
- JSCDC IRB: Hui-Yuan Cai, Tel: +86-25-83759406; Fax: +86-25-83759406
- DSMB: Jie-Lai Xia, Tel: 029-84774858; Fax: 029-84774858
The unblinding of single cases by investigators in the course of the clinical trial will only be performed if relevant for the safety of the participant.
In addition to the initial 24-hour report, a completed, separate SAE report is to be sent to the sponsors, principle investigator, JSCDC IRB, and the DSMB via fax or mail within 48 hours of the event as following report, and an end report after the resolve of the SAE. All SAEs will be recorded on the case report form or other source documents.
It is the responsibility of the sponsor to ensure that all information pertaining to suspected unexpected serious adverse reactions (SUSARs) due to an investigational medicine in a clinical trial.
The sponsor is responsible for the prompt notification to all concerned investigators, the JSCDC IRB and competent authority of all relevant safety information previously described.
Fatal or life-threatening SUSARs will be reported to the competent authority and the JSCDC IRB as soon as possible but no later than seven calendar days after the sponsor has first knowledge of the minimum criteria for expedited reporting.
In each case relevant follow-up information will be sought and a report completed as soon as possible. It will be communicated to the competent authority and the JSCDC IRB within an additional eight calendar days.
All other SUSARs and safety issues deserving expedited reporting must be reported to the competent authority and dependent on national provisions to the JSCDC IRB in the concerned countries as soon as possible but no later than 15 calendar days after the sponsor has first knowledge of the minimum criteria for expedited reporting.
Expedited reporting is not usually required for reactions which are serious but expected, or for non-serious adverse reactions whether expected or not.
Safety data monitoring by DSMB
The DSMB will blindly review safety continuously during the trial and will advise on progression of the trial. Table S6 provides the members of the data and safety monitoring board. It will review the safety data from the trial as generated by the investigators, in order to advise whether or not the study integrity remains intact and whether or not there are any safety concerns worth early notice. DSMB will review the reported safety data in the participants for the first 7 days after vaccination. During the study period, if an increase of risk for participants is noticed, the DSMB should promptly inform the principle investigator and sponsors. Sponsors, investigators and DSMB will have a panel meeting, and then DSMB will make final decision to pause or all an early termination of the study.
Administration of study injections and new enrollments will be paused, if:
- One serious adverse event may be associated with vaccination, or
- Occurrence of grade 3 adverse reaction may be associated with vaccination in 10% of participants or more (including injection-site reaction, systemic reaction, and change of the safety laboratory measures).
The study may come to an early termination, if:
- One vaccination-associated serious adverse event, or
- Occurrence of grade 3 adverse events associated with vaccination in 15% of participants or more (including injection-site reaction, systemic reaction, and change of the safety laboratory measures), or
- Required by sponsor, or
- Required by regulatory authority, or
- Required by institutional review board (IRB).
Withdrawal due to adverse events (see also section on participant withdrawal)
Withdrawal due to AE should be distinguished from withdrawal due to other reasons unrelated to safety issues, according to the definition of AE noted earlier and recorded on the appropriate AE CRF page.
When a participant withdraws due to an SAE, the SAE must be reported in accordance with the reporting requirements defined above.
Laboratory assay and immunogenicity assessments
Antigen-specific antibody responses
Antigen-specific antibodies response will be determined at day 0 (immediately before vaccination), day 7, day 14, day 21, day 42, month 3 and month 6. The antibodies responses in different time points will be compared between treatment groups. ELISA will be applied for the detection of the antigen-specific antibodies responses.
Cellular immunity responses
Peripheral blood mononuclear cells (PBMC) will be isolated from venous blood and stimulated with overlapping peptide pools of GP protein for 48 hours. The cellular immunity responses will be tested at day 0 (immediately before vaccination), day 7, day 14, day 21, day 42, month 3 and month 6 by enzyme-linked immunospot assay (ELISPOT).
Functional antibody level
Functional antibody responses will be detected at day 0 (immediately before vaccination), day 7, day 14, day 21, day 42, month 3 and month 6. The functional antibody level pre-vaccination and the post-vaccination will be compared to reveal the difference. And the functional antibody level at different time will be compared to reveal the difference.
Nasal sampling swab
Nasal sampling swab will be detected for S. aureus colonization on nasal mucosa at day 0, day 3, day 7, day 10, day 14, day 17, day 21, day 42, month 3 and month 6.
Antigen-specific antibody responses
Antigen-specific antibodies response will be determined at day 0 (immediately before vaccination), day 7, day 14, day 21, day 42, month 3 and month 6. The antibodies responses in different time points will be compared between treatment groups. ELISA will be applied for the detection of the antigen-specific antibodies responses.
Cellular immunity responses
Peripheral blood mononuclear cells (PBMC) will be isolated from venous blood and stimulated with overlapping peptide pools of GP protein for 48 hours. The cellular immunity responses will be tested at day 0 (immediately before vaccination), day 7, day 14, day 21, day 42, month 3 and month 6 by enzyme-linked immunospot assay (ELISPOT).
Functional antibody level
Functional antibody responses will be detected at day 0 (immediately before vaccination), day 7, day 14, day 21, day 42, month 3 and month 6. The functional antibody level pre-vaccination and the post-vaccination will be compared to reveal the difference. And the functional antibody level at different time will be compared to reveal the difference.
Nasal sampling swab
Nasal sampling swab will be detected for S. aureus colonization on nasal mucosa at day 0, day 3, day 7, day 10, day 14, day 17, day 21, day 42, month 3 and month 6.
Data collection and management
Source documents and source data
The purpose of source documents is to document the existence of the participant and substantiate the integrity of the trial data collected. The Investigator must maintain the trial source documents accurate, complete, legible and up to date.
Examples of source documents are: participant screening, laboratory measure reports, enrolment log, participant’s diary cards, hospital records, informed consent forms, investigational dispensing and reconciliation forms, participant’s file and records kept at the pharmacy or at the laboratories, mail, certified letters.
Source data are the data contained in source documents (originals or certified copies). The investigator is responsible for the accuracy and completeness of the data reported in source documents. Data reported in the case report forms (CRFs) that are derived from source documents should be consistent with source documents and any discrepancies should be explained.
All CRFs must be signed by the Investigator. Incorrect data must be crossed-out with a single line, then initialed and dated. Correction fluid or similar corrective methods that mask the original data will not to be used. These rules also apply to the completion of SAE Reporting Forms, Data Correction Forms, and ICFs.
Clinical data management
CRF is used to record data in clinical trials, is an important part of clinical trials and research reports, filling shall be clear and intact, and also should be completed with Chinese language and black pen. Only authorized investigators could correct the errors in the CRFs, The original record can’t be obliterated or overwrite. Investigator should draw a horizontal line across the original data which should be corrected, and specify the corrected data in the space next to them, and noted the signature and date.
According to the project requirements, the data collection, biological sample collection and examination should be done in the visit window, the original documents and records shall be complete. The result of the examination also should be timely entered into case report form (CRF).
Auditors should conduct regular and irregular audits of data records until CRF are completed, auditors should carefully verify CRF number of the participants, the number of pages in each CRF and necessary signatures of researchers. The main contents of audits should be focused on signed informed consent, volunteer screening into the group, vaccination, management of the investigational vaccine, safety observation and immunogenicity of specimen collection and preservation, Consistency between research data and the original data is the emphasis of audits. Manual verification results shall be recorded. Transfer of CRF research data should be documented. For each batch of data, double entry, quality control and triggers to computerized logic and/or consistency checks will be systematically applied in order to detect errors or omissions. Queries will be generated and submitted through Data Clarification Forms to the Investigator for resolution.
SAE data management
During the trial, all the SAEs will be reported on SAE Reporting Forms and integrated into the Sponsor’s SAE database. Upon receipt of an SAE Reporting Form, the data will be entered into the database after a duplicate check. Each SAE is assigned a case identification number. Entered data will be independently verified against the original SAE forms. All SAEs are then reviewed by the DSMB. Each SAE is reviewed, locked and approved in the database. Any follow-up information concerning a locked and approved SAE will be incorporated and a new version of the SAE will be created.
Archiving
Following closure of the study, the investigator must maintain all site study records in a safe and secure location. The records must be maintained to allow easy and timely retrieval, when needed (e.g., audit or inspection), and, whenever feasible, to allow any subsequent review of data in conjunction with assessment of the facility, supporting systems, and staff. Where permitted by applicable laws/regulations or institutional policy, some or all of these records can be maintained in a validated format other than hard copy; however, caution needs to be exercised before such action is taken. The investigator must assure that all reproductions are legible and are a true and accurate copy of the original ones, and meet accessibility and retrieval standards, including re-generating a hard copy, if required. Furthermore, the investigator must ensure there is an acceptable back-up of these reproductions and that an acceptable quality control process exists for making these reproductions.
Trial-related documents will be maintained for a period of 10 years after final marketing approval of the vaccine, or 10 years after the formal discontinuation of clinical development of the product per the requirements by SFDA and IRB/IEC. The site investigators must be aware of all requirements and retain protocol records in accordance with the longest requirement that pertains to the study. No study document should be destroyed without prior written agreement between the sponsors and the principal investigator. Storage of all trial-related documents will be such that confidentiality will be strictly maintained.
Database creation and data entry
The person, who is responsible for data management, establish the database structure and inspection procedures to ensure that the database can be correctly converted to SAS file format, and the database structure can also be modified and confirmed by tentative data entry.
Before CRF entry, data management staff will carry out the CRF verification again, mainly to see if there are obvious errors and omissions. Data entry staff will start data entry after several training, double entry will be accepted.
The consistency check of the database that be independently completed by two persons will be executed. Inconsistent values and information should be reported. After that, the raw data will be checked one by one and the mistake will be corrected until database is left in a consistent state. The computer program that has been prepared and confirmed will be applied to do logical consistency check. Before modifying the database, Query table should be shown and confirmed by researchers until there is no question. A certain proportion of CRF will be randomly selected to finish quality control, and to compare with the data in the database, to ensure that the data in the database and the CRF content are consistent.
The database lock
Blind review of the database is required before Statistical analysis. The aim of blind review is to determine the population that will be analyzed according the evaluation criteria, including full analysis set (FAS) under the Principle of intention to treat (ITT) analysis, per-protocol set (PPS) and safety analyzes data set (SS), confirmation of the deviation from the project and other influences on database. Database will be locked after blind review be confirmed.
Source documents and source data
The purpose of source documents is to document the existence of the participant and substantiate the integrity of the trial data collected. The Investigator must maintain the trial source documents accurate, complete, legible and up to date.
Examples of source documents are: participant screening, laboratory measure reports, enrolment log, participant’s diary cards, hospital records, informed consent forms, investigational dispensing and reconciliation forms, participant’s file and records kept at the pharmacy or at the laboratories, mail, certified letters.
Source data are the data contained in source documents (originals or certified copies). The investigator is responsible for the accuracy and completeness of the data reported in source documents. Data reported in the case report forms (CRFs) that are derived from source documents should be consistent with source documents and any discrepancies should be explained.
All CRFs must be signed by the Investigator. Incorrect data must be crossed-out with a single line, then initialed and dated. Correction fluid or similar corrective methods that mask the original data will not to be used. These rules also apply to the completion of SAE Reporting Forms, Data Correction Forms, and ICFs.
Clinical data management
CRF is used to record data in clinical trials, is an important part of clinical trials and research reports, filling shall be clear and intact, and also should be completed with Chinese language and black pen. Only authorized investigators could correct the errors in the CRFs, The original record can’t be obliterated or overwrite. Investigator should draw a horizontal line across the original data which should be corrected, and specify the corrected data in the space next to them, and noted the signature and date.
According to the project requirements, the data collection, biological sample collection and examination should be done in the visit window, the original documents and records shall be complete. The result of the examination also should be timely entered into case report form (CRF).
Auditors should conduct regular and irregular audits of data records until CRF are completed, auditors should carefully verify CRF number of the participants, the number of pages in each CRF and necessary signatures of researchers. The main contents of audits should be focused on signed informed consent, volunteer screening into the group, vaccination, management of the investigational vaccine, safety observation and immunogenicity of specimen collection and preservation, Consistency between research data and the original data is the emphasis of audits. Manual verification results shall be recorded. Transfer of CRF research data should be documented. For each batch of data, double entry, quality control and triggers to computerized logic and/or consistency checks will be systematically applied in order to detect errors or omissions. Queries will be generated and submitted through Data Clarification Forms to the Investigator for resolution.
SAE data management
During the trial, all the SAEs will be reported on SAE Reporting Forms and integrated into the Sponsor’s SAE database. Upon receipt of an SAE Reporting Form, the data will be entered into the database after a duplicate check. Each SAE is assigned a case identification number. Entered data will be independently verified against the original SAE forms. All SAEs are then reviewed by the DSMB. Each SAE is reviewed, locked and approved in the database. Any follow-up information concerning a locked and approved SAE will be incorporated and a new version of the SAE will be created.
Archiving
Following closure of the study, the investigator must maintain all site study records in a safe and secure location. The records must be maintained to allow easy and timely retrieval, when needed (e.g., audit or inspection), and, whenever feasible, to allow any subsequent review of data in conjunction with assessment of the facility, supporting systems, and staff. Where permitted by applicable laws/regulations or institutional policy, some or all of these records can be maintained in a validated format other than hard copy; however, caution needs to be exercised before such action is taken. The investigator must assure that all reproductions are legible and are a true and accurate copy of the original ones, and meet accessibility and retrieval standards, including re-generating a hard copy, if required. Furthermore, the investigator must ensure there is an acceptable back-up of these reproductions and that an acceptable quality control process exists for making these reproductions.
Trial-related documents will be maintained for a period of 10 years after final marketing approval of the vaccine, or 10 years after the formal discontinuation of clinical development of the product per the requirements by SFDA and IRB/IEC. The site investigators must be aware of all requirements and retain protocol records in accordance with the longest requirement that pertains to the study. No study document should be destroyed without prior written agreement between the sponsors and the principal investigator. Storage of all trial-related documents will be such that confidentiality will be strictly maintained.
Database creation and data entry
The person, who is responsible for data management, establish the database structure and inspection procedures to ensure that the database can be correctly converted to SAS file format, and the database structure can also be modified and confirmed by tentative data entry.
Before CRF entry, data management staff will carry out the CRF verification again, mainly to see if there are obvious errors and omissions. Data entry staff will start data entry after several training, double entry will be accepted.
The consistency check of the database that be independently completed by two persons will be executed. Inconsistent values and information should be reported. After that, the raw data will be checked one by one and the mistake will be corrected until database is left in a consistent state. The computer program that has been prepared and confirmed will be applied to do logical consistency check. Before modifying the database, Query table should be shown and confirmed by researchers until there is no question. A certain proportion of CRF will be randomly selected to finish quality control, and to compare with the data in the database, to ensure that the data in the database and the CRF content are consistent.
The database lock
Blind review of the database is required before Statistical analysis. The aim of blind review is to determine the population that will be analyzed according the evaluation criteria, including full analysis set (FAS) under the Principle of intention to treat (ITT) analysis, per-protocol set (PPS) and safety analyzes data set (SS), confirmation of the deviation from the project and other influences on database. Database will be locked after blind review be confirmed.
Statistics plan and statistical analysis
Interim statistical analysis
Interim analyses will be conducted by an independent statistical party after the last participant completes Visit 8 on day 42. The data collected from day 0 to day 42 will be unblinded by representatives of statistical party, sponsor, clinical research investigator and Clinical Research Organization (CRO). The un-blinding data will not be disclosed to the participants, staff who perform the site works and follow-up, and the laboratory staff who test the bio-samples. Therefore the clinical trial site will still be kept in blind. The interim results will not influence the later safety or immunogenicity endpoint assessments.
The final analysis
Final analyses will be conducted by a statistical party, after the last participant completes Visit 10 on 6 month, and all the safety data and immunogenicity data collected after day 42 up to month 6 will be reviewed and then locked for the final statistical analysis.
Analyzed data sets definition
All randomized participants who received vaccination should be included in the safety evaluation. Thus, the safety analysis cohort will be determined on the basis of (Intention-To-Treat) ITT cohort, including all participants received at least one shot.
Events will be reported on per-individual basis, i.e., counting individuals rather than events. This means that even if a participant suffered a same event repeatedly during the follow-up, the event will be counted only once, except for SAEs. Repeated same adverse events in participant will be summarized according to the following rule: if a participant suffered the same adverse event more than once, the event will be assigned the worst severity, the closest relationship to the vaccination and the earliest starting date. In the listings, however, all occurrences of the adverse events will be shown.
Full analysis set (FAS) for immunogenicity analysis: FAS is determined based on ITT, including all of the participants that meet the inclusion/exclusion criteria, randomization, receiving vaccination, and have at least one blood testing result.
Per-protocol set (PPS): it is a subset of FAS. In this set, all participants that meet the inclusion/exclusion criteria and complete the vaccine inoculation within visit window according to the protocol, and complete the blood collection at all scheduled visits, with no significant deviation or violation of protocol.
In this study, the PPS are the primary analysis set for immunogenicity evaluation, but the FAS will also be analyzed at the same time. Any difference of analysis results existed between PPS and FAS, will be discussed in the report.
Statistical methods
Safety analysis of this experiment is mainly descriptive analysis of incidence rate of adverse reaction or adverse events. A chi-square test can be used to compare the proportion of groups, Fisher’s exact test will be used when it is necessary. Analysis of immunogenicity indicators on antibody levels need to do logarithmic transformation, the results of analysis should be shown in GMT, standard deviation, median, minimum and maximum values and 95% confidence intervals, chi-square test can be used to compare categorical indicators between groups such as positive conversion rate of immune response, if it is necessary, Fisher's exact test will be used. Statistical analysis method of repeated measures data can be used to analyze experimental data at different time points in this study.
SAS (version 9.3) was used for all analyses, test statistics and the corresponding p values are given. All statistical tests were two-sided and significance was set at P≤0.05 (more detailed information, please read the reference, the interim-term statistical analysis plan and the final statistical analysis plan).
Interim statistical analysis
Interim analyses will be conducted by an independent statistical party after the last participant completes Visit 8 on day 42. The data collected from day 0 to day 42 will be unblinded by representatives of statistical party, sponsor, clinical research investigator and Clinical Research Organization (CRO). The un-blinding data will not be disclosed to the participants, staff who perform the site works and follow-up, and the laboratory staff who test the bio-samples. Therefore the clinical trial site will still be kept in blind. The interim results will not influence the later safety or immunogenicity endpoint assessments.
The final analysis
Final analyses will be conducted by a statistical party, after the last participant completes Visit 10 on 6 month, and all the safety data and immunogenicity data collected after day 42 up to month 6 will be reviewed and then locked for the final statistical analysis.
Analyzed data sets definition
All randomized participants who received vaccination should be included in the safety evaluation. Thus, the safety analysis cohort will be determined on the basis of (Intention-To-Treat) ITT cohort, including all participants received at least one shot.
Events will be reported on per-individual basis, i.e., counting individuals rather than events. This means that even if a participant suffered a same event repeatedly during the follow-up, the event will be counted only once, except for SAEs. Repeated same adverse events in participant will be summarized according to the following rule: if a participant suffered the same adverse event more than once, the event will be assigned the worst severity, the closest relationship to the vaccination and the earliest starting date. In the listings, however, all occurrences of the adverse events will be shown.
Full analysis set (FAS) for immunogenicity analysis: FAS is determined based on ITT, including all of the participants that meet the inclusion/exclusion criteria, randomization, receiving vaccination, and have at least one blood testing result.
Per-protocol set (PPS): it is a subset of FAS. In this set, all participants that meet the inclusion/exclusion criteria and complete the vaccine inoculation within visit window according to the protocol, and complete the blood collection at all scheduled visits, with no significant deviation or violation of protocol.
In this study, the PPS are the primary analysis set for immunogenicity evaluation, but the FAS will also be analyzed at the same time. Any difference of analysis results existed between PPS and FAS, will be discussed in the report.
Statistical methods
Safety analysis of this experiment is mainly descriptive analysis of incidence rate of adverse reaction or adverse events. A chi-square test can be used to compare the proportion of groups, Fisher’s exact test will be used when it is necessary. Analysis of immunogenicity indicators on antibody levels need to do logarithmic transformation, the results of analysis should be shown in GMT, standard deviation, median, minimum and maximum values and 95% confidence intervals, chi-square test can be used to compare categorical indicators between groups such as positive conversion rate of immune response, if it is necessary, Fisher's exact test will be used. Statistical analysis method of repeated measures data can be used to analyze experimental data at different time points in this study.
SAS (version 9.3) was used for all analyses, test statistics and the corresponding p values are given. All statistical tests were two-sided and significance was set at P≤0.05 (more detailed information, please read the reference, the interim-term statistical analysis plan and the final statistical analysis plan).
Ethical and legal issues
Guideline
The study will be conducted according to GCP, the Declaration of Helsinki, and local rules and regulations of China. Submission of the protocol and any protocol amendments to regulatory agencies will occur in accordance with local regulatory requirements. When submission to the local regulatory authority is required, the timing of the submission relative to IRB submission or approval and whether or not the authority will provide their approval of or favorable opinion on the protocol or amendment before it can be implemented will depend on local regulatory requirements.
Institutional Review Board
The investigator is responsible for obtaining written approval for the clinical study protocol (including all substantial protocol amendments), the written participant informed consent form, informed consent updates, participant recruitment procedures (e.g., advertisements) and any other written information to be provided to participants from an IRB which complies with local regulatory requirements. Any amendments will require approval by the IRB.
The only circumstance in which an amendment may be initiated prior to JSCDC IRB approval is where the change is necessary to eliminate apparent immediate hazards to the participants. In that event, the investigator must notify the Institutional Review Board and the sponsor in writing immediately after the implementation.
A final study notification will be forwarded by the investigator to the JSCDC IRB within 90 days after the study has been completed or in the event of premature termination of the study within 15 days. Copies of all clinical study status reports (including termination) will be provided by an investigator to CRO.
Ethical conduct of the study
- To ensure that he/she has sufficient time to conduct and complete the study and has adequate staff and appropriate facilities and equipment which are available for the duration of the study and to ensure that other studies do not divert essential participants or facilities away from the study at hand.
- To submit an up-to-date curriculum vitae or Investigator Biography and other credentials to the sponsors and where required to relevant authorities. It is recommended that this documentation indicates any previous clinical research experience and history of training in GCP.
- To acquire the reference ranges for laboratory tests performed locally and, if required by local regulations, obtain the laboratory’s current certification or Quality Assurance procedure manual.
- To ensure that all back-up clinical samples (including serum samples) are retained onsite according to the approval of the sponsors.
- To perform no other biological assays on the clinical samples except those described in the protocol or its amendment(s).
- To prepare and maintain adequate source data or raw data designed to record observations, and other data pertinent to the study.
- To conduct the study in compliance with the protocol any amendment and “Good Clinical Practice” (GCP) and all applicable regulatory requirements.
- To cooperate with a representative of the sponsors in the monitoring process of the study and in resolution of queries about the data.
- To permit drug regulatory agencies and sponsors’ audits.
Protocol amendments and administrative changes
No changes to the study protocol will be allowed unless approved by IRB. This does not apply to changes made to reduce discomfort or avert risk to study participants. Furthermore, in the event of a medical emergency, the investigators shall perform any medical procedures that are deemed medically appropriate. The principle investigator must notify the sponsor of all such occurrences.
Written IRB approval of protocol amendments is required prior to implementation, except where permitted by all applicable regulatory requirements; administrative changes and amendments not submitted for approval are submitted to IRB for information only. Any amendment/administrative change to the protocol will be adhered to by the participating center(s) and will apply to all participants. Submission of protocol amendments to regulatory agencies will occur in accordance with local regulatory requirements. When submission to the local regulatory authority is required, the timing of the submission relative to IRB submission or approval and whether or not the authority will provide their approval of or favorable opinion on the amendment before it can be implemented will depend on local regulatory requirements.
Confidentiality of data and access to participant records
Prior to initiation of the trial, the investigators will sign a fully executed confidentiality agreement with the sponsor. All study-related information will be stored securely at the study sites. All participant information will be stored in locked file cabinets in areas with access limited to study staff. All laboratory specimens, reports, study data collection, process, and administrative forms will be identified by coded number only to maintain participant confidentiality. All computer entries will be done by coded numbers only, and all local databases will be secured with password-protected access systems. Forms, lists, logbooks, appointment books, and any other listings that link participant ID numbers to other identifying information will be stored in a separate, locked file in an area with limited access. Sponsor personnel, the IRB and the regulatory authorities will have direct access to source data/documents.
Guideline
The study will be conducted according to GCP, the Declaration of Helsinki, and local rules and regulations of China. Submission of the protocol and any protocol amendments to regulatory agencies will occur in accordance with local regulatory requirements. When submission to the local regulatory authority is required, the timing of the submission relative to IRB submission or approval and whether or not the authority will provide their approval of or favorable opinion on the protocol or amendment before it can be implemented will depend on local regulatory requirements.
Institutional Review Board
The investigator is responsible for obtaining written approval for the clinical study protocol (including all substantial protocol amendments), the written participant informed consent form, informed consent updates, participant recruitment procedures (e.g., advertisements) and any other written information to be provided to participants from an IRB which complies with local regulatory requirements. Any amendments will require approval by the IRB.
The only circumstance in which an amendment may be initiated prior to JSCDC IRB approval is where the change is necessary to eliminate apparent immediate hazards to the participants. In that event, the investigator must notify the Institutional Review Board and the sponsor in writing immediately after the implementation.
A final study notification will be forwarded by the investigator to the JSCDC IRB within 90 days after the study has been completed or in the event of premature termination of the study within 15 days. Copies of all clinical study status reports (including termination) will be provided by an investigator to CRO.
Ethical conduct of the study
- To ensure that he/she has sufficient time to conduct and complete the study and has adequate staff and appropriate facilities and equipment which are available for the duration of the study and to ensure that other studies do not divert essential participants or facilities away from the study at hand.
- To submit an up-to-date curriculum vitae or Investigator Biography and other credentials to the sponsors and where required to relevant authorities. It is recommended that this documentation indicates any previous clinical research experience and history of training in GCP.
- To acquire the reference ranges for laboratory tests performed locally and, if required by local regulations, obtain the laboratory’s current certification or Quality Assurance procedure manual.
- To ensure that all back-up clinical samples (including serum samples) are retained onsite according to the approval of the sponsors.
- To perform no other biological assays on the clinical samples except those described in the protocol or its amendment(s).
- To prepare and maintain adequate source data or raw data designed to record observations, and other data pertinent to the study.
- To conduct the study in compliance with the protocol any amendment and “Good Clinical Practice” (GCP) and all applicable regulatory requirements.
- To cooperate with a representative of the sponsors in the monitoring process of the study and in resolution of queries about the data.
- To permit drug regulatory agencies and sponsors’ audits.
Protocol amendments and administrative changes
No changes to the study protocol will be allowed unless approved by IRB. This does not apply to changes made to reduce discomfort or avert risk to study participants. Furthermore, in the event of a medical emergency, the investigators shall perform any medical procedures that are deemed medically appropriate. The principle investigator must notify the sponsor of all such occurrences.
Written IRB approval of protocol amendments is required prior to implementation, except where permitted by all applicable regulatory requirements; administrative changes and amendments not submitted for approval are submitted to IRB for information only. Any amendment/administrative change to the protocol will be adhered to by the participating center(s) and will apply to all participants. Submission of protocol amendments to regulatory agencies will occur in accordance with local regulatory requirements. When submission to the local regulatory authority is required, the timing of the submission relative to IRB submission or approval and whether or not the authority will provide their approval of or favorable opinion on the amendment before it can be implemented will depend on local regulatory requirements.
Confidentiality of data and access to participant records
Prior to initiation of the trial, the investigators will sign a fully executed confidentiality agreement with the sponsor. All study-related information will be stored securely at the study sites. All participant information will be stored in locked file cabinets in areas with access limited to study staff. All laboratory specimens, reports, study data collection, process, and administrative forms will be identified by coded number only to maintain participant confidentiality. All computer entries will be done by coded numbers only, and all local databases will be secured with password-protected access systems. Forms, lists, logbooks, appointment books, and any other listings that link participant ID numbers to other identifying information will be stored in a separate, locked file in an area with limited access. Sponsor personnel, the IRB and the regulatory authorities will have direct access to source data/documents.
Financial contract and insurance coverage
An agreement will be signed by all the parties involved in the trial’s performance, if relevant. Adequate insurance coverage for all participants to be included in the trial is supplied by the sponsor.
Publication of study results
Sponsors have no objection to publication by investigator of any information collected or generated by investigator, whether or not the results are favorable to the investigational product. However, to ensure against inadvertent disclosure of Confidential Information or unprotected inventions, investigator will provide sponsors an opportunity to review any proposed publication or other type of disclosure before it is submitted or otherwise disclosed.
Table S1
| Vaccine | Antigen target | Company | Clinical trial phase |
|---|---|---|---|
| V710 | Iron surface determinant B (IsdB) | Merck/Intercell | Phase 3 |
| StaphVax | CPS types5+8 | Nabi Biota pharmaceuticals | Phase 3 |
| Bivalent rLukS-PV/rAT | rLukS-PV/rAT | Nabi Biota pharmaceuticals | Phase 2 |
| SA3Ag | CP5\CP8\ClfA | Pfizer | Phase 2 |
| SA4Ag | CP5\CP8\ClfA\MntC | Pfizer | Phase 2 |
| NDV-3 | Candida Als3 | Novartis | Phase 2 |
| GSK2392103A | Recombinant proteins | GSK | Phase 1 |
| GSK2392105A | |||
| GSK2392106A | |||
| GSK2392019A | |||
| STEBVax | Staphylococcal enterotoxin B | NIAID | Phase 1 |
Table S2
| Visit (V) | Scheduled time point for visit | Time window |
|---|---|---|
| Visit 0 (V0) | Within 7 days before vaccination | – |
| Visit 1 (V1) | The first vaccination at day 0 | – |
| Visit 2 (V2) | Day 3 after the first vaccination | +1 day |
| Visit 3 (V3) | Day 7 after the first vaccination | +1 day |
| Visit 4 (V4) | Day 10 after the first vaccination (3 days after V3) | ±1 day |
| Visit 5 (V5) | Day 14 after the first vaccination | ±1 day |
| Visit 6 (V6) | Day 17 after the first vaccination (3 days after V5) | ±1 day |
| Visit 7 (V7) | Day 21 after the first vaccination (7 days after V5) | +2 days |
| Visit 8 (V8) | Day 42 after the first vaccination (28 days after V5) | +3 days |
| Visit 9 (V9) | Month 3 after the first vaccination | ±7 days |
| Visit 10 (V10) | Month 6 after the first vaccination | ±7 days |
Table S3
| Activities | V0 | V1 | V2 | V3 | V4 | V5 | V6 | V7 | V8 | V9 | V10 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Within 7 days before day 0 | D0 | D3 | D7 | D10 | D14 | D17 | D21 | D42 | M3 | M6 | |
| Informed consent | ☆ | ||||||||||
| Collecting demographic information | ☆ | ||||||||||
| Preliminarily check inclusion and exclusion criteria | ☆ | ||||||||||
| Physical examination: | |||||||||||
| Height, weight, BMI | ☆ | ||||||||||
| Respiratory rate, pulse rate, blood pressure | ☆ | ||||||||||
| Urine pregnancy test (female) | ☆ | ||||||||||
| Routine blood test | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |||||
| Blood biochemical test | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |||||
| Pre-vaccination body temperature | ☆ | ☆ | ☆ | ☆ | |||||||
| Inclusion and exclusion criteria before enrolled | ☆ | ||||||||||
| Follow-up vaccination exclusion criteria | ☆ | ☆ | ☆ | ||||||||
| Randomization | ☆ | ||||||||||
| Serum analyses: | |||||||||||
| Five antigen-specific antibody response by ELISA | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ||||
| Functional antibody level | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ||||
| Cellular immune responses | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ||||
| Nasal sampling swab | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |
| Vaccination | ☆ | ☆ | ☆ | ☆ | |||||||
| Observation for 30 minutes post-vaccination, participates safety observation training | ☆ | ☆ | ☆ | ☆ | |||||||
| Distribution of diary cards, ruler, signing-pen, thermometer | ☆ | ☆ | ☆ | ||||||||
| Record on the “Vaccination and visits record” | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |
| Participates safety observations self-report in diary cards | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |||
| Report serious adverse events | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |
| Return of diary cards | ☆ | ☆ | ☆ |
Table S4
| Activities | V0 | V1 | V2 | V3 | V4 | V5 | V6 | V7 | V8 | V9 | V10 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Within 7 days before day 0 | Day0 | Day3 | Day7 | Day10 | Day14 | Day17 | Day21 | Day42 | Mon 3 | Mon6 | |
| Clinical evaluations | |||||||||||
| Routine blood test | 2 | – | 2 | 2 | 2 | 2 | 2 | – | – | – | – |
| Blood biochemical test | 5 | – | 5 | 5 | 5 | 5 | 5 | – | – | – | – |
| Immunogenicity | |||||||||||
| Antibody assays and serum storage | – | 8 | – | 8 | 8 | 8 | 8 | 8 | 8 | ||
| PBMC and plasma for storage | – | 16 | – | 16 | 16 | 16 | 16 | 16 | 16 | ||
| Daily volume | 7 | 24 | 7 | 31 | 7 | 31 | 7 | 24 | 24 | 24 | 24 |
| Cumulative volume | 7 | 31 | 38 | 69 | 76 | 107 | 114 | 138 | 162 | 186 | 210 |
Table S5
| Laboratory indexes | Mild (grade 1) | Moderate (grade 2) | Severe (grade 3) | Potentially life threatening (grade 4) |
|---|---|---|---|---|
| Hemoglobin (female) change from baseline value, gm/dL | Any increase –1.5 | 1.6–2.0 | 2.1–5.0 | >5.0 |
| Hemoglobin (male) change from baseline value, gm/dL | Any increase –1.5 | 1.6–2.0 | 2.1–5.0 | >5.0 |
| Hemoglobin (female), gm/dL | 12.0–13.0 | 10.0–11.9 | 8.0–9.9 | <8.0 |
| Hemoglobin (male), gm/dL | 12.5–14.5 | 10.5–12.4 | 8.5–10.4 | <8.5 |
| WBC increase, cell/mm3 | >13,000/mm3 (>13×109/L) | 13,000–15,000/mm3 (13–15×109/L) | 15,000–30,000/mm3 (15–30×109/L) | >30,000/mm3 (>30×109/L) |
| WBC decrease, cell/mm3 | 2,500–3,500/mm3 (2.5–3.5×109/L) | 1,500–2,499/mm3 (1.5–2.499×109/L) | 1,000–1,499/mm3 (1.0–1.499×109/L) | <1,000/mm3 |
| Liver function tests—ALT, AST increase by factor | 1.25–2.5× ULN* | 2.6–5× ULN | 5.1–10× ULN | >10× ULN |
| Creatinine increase by factor | 1.1–1.5× ULN | 1.6–3.0× ULN | 3.1–6× ULN | >6× ULN |
*ULN, is the upper limit of the normal range.
Table S6
| Name | Title | Work units | Major | Position |
|---|---|---|---|---|
| Jie-Lai Xia | Professor | Public Health Department, Fourth Military Medical University | Statistics | Chairman |
| Xuan-Yi Wang | Professor | Institute of Biomedical Sciences Fudan University | Epidemiology | Member |
| Tao Wang | Professor | Institute of immunology, Second Military Medical University | Immunology | Member |
| Xiang-Dong Zhou | Professor of Medicine | Southwest Hospital | Clinical infection disease | Member |
| Hui-Wang | Professor of Medicine | Peking People’s Hospital | Clinical infection disease | Member |
| Brief Title: | A phase 1 clinical trial of a recombinant staphylococcus aureus vaccine (RSAV) in healthy adults |
|---|---|
| Protocol Title: | A single-center, randomized, double-blind, placebo-controlled clinical trial to evaluate the safety, tolerability and preliminary immunogenicity of a recombinant staphylococcus aureus vaccine (RSAV) in healthy adults aged between 18 and 65 years in China |
| Protocol Number: | JSVCT027 |
| ClinicalTrials.gov ID: | NCT02804711 |
| Protocol Date: | 5-Sep-16 |
| Version: | Version 1.4 (final) |
| Phase: | Phase 1 |
| Sponsor: | Olymvax Biopharmaceuticals Inc.; Third Military Medical University |
| Investigational vaccines: | Recombinant staphylococcus aureus vaccine (RSAV) |
| Principle Investigator | Feng-Cai Zhu, MSc |
| Protocol Authors | Feng-Cai Zhu; Jiangsu Provincial Center for Disease Control and Prevention |
| Quan-Ming Zou; Third Military Medical University | |
| Ming Zeng; National Institutes for Food and Drug Control | |
| Hao Zeng; Third Military Medical University | |
| Bin Wang; National Institutes for Food and Drug Control | |
| Yue-Mei Hu; Jiangsu Provincial Center for Disease Control and Prevention | |
| Fan-Yue Meng; Jiangsu Provincial Center for Disease Control and Prevention | |
| Jing-Xin Li; Jiangsu Provincial Center for Disease Control and Prevention | |
| Jiang Gu; Third Military Medical University | |
| Liu-Sheng Peng; Third Military Medical University |
This document contains confidential information belonging to Olymvax Biopharmaceuticals Inc. and Third Military Medical University.
| Document history | ||
|---|---|---|
| Version No. | Version date | Amendment |
| Sponsor | Chengdu Olymvax Biopharmaceuticals Inc. No. 99, Tianxin Street, Gaoxin District, Chengdu 611731, China |
| Third Military Medical University, No. 30 Gaotanyan Street, Shapingba District, Chongqing 400038, China | |
| Tel: 028-69361119 | |
| Fax: 028-69361100 | |
| Email: saowen@vip.163.com | |
| Principal investigator | Feng-Cai Zhu; Jiangsu Provincial Center for Diseases Control and Prevention, No. 172, Jiangsu Road, Nanjing 210009, Jiangsu Province, China |
| Tel: +86-25-83759418 | |
| Fax: +86-25-83759409 | |
| Email: jszfc@vip.sina.com | |
| Contract Research Organization | Yuan Yong; Shenzhen Yingheyuan Medical Technology Development Co., Ltd., 6B-01, Tian-Xia Industrial Park, Ma Jia Long, Yi-Yuan Road, Nan-Shan District, Shenzhen 518052, China |
| Tel: +86-755-61373860 | |
| Fax: +86-755-61373008 | |
| Email: 13382030658@189.cn | |
| The Data and Safety Monitoring Committee | Jie-Lai Xia; Public Health Department The Fourth Military Medical University, No. 169 west of Changle Road, Xi’an 710032, Shanxi Province, China |
| Tel: 029-84774858 | |
| Fax: 029-84774858 | |
| Email: jielaixia@yahoo.com | |
| Clinical Laboratory | Ming Zeng; National Institute for Food and Drug Control, No. 2 west of Tiantan Road, Beijing 100050, China |
| Tel: 010-67058402 | |
| Fax: 010-67058402 | |
| Email: zengming@nicpbp.org.cn | |
| Statistical Party | Pei Liu; Public Health Department, Southeast University, No. 78 Dingjiaqiao, Nanjing 210009, Jiangsu Province, China |
| Tel: +86-25-83272584 | |
| Fax: +86-25-83272584 | |
| Email: liupeiseu@126.com |
| Brief Title | A phase 1 clinical trial of a recombinant staphylococcus aureus vaccine (RSAV) in healthy adults |
|---|---|
| Official Title | A single-center, randomized, double-blind, placebo-controlled clinical trial to evaluate the safety, tolerability and preliminary immunogenicity of a recombinant staphylococcus aureus vaccine (RSAV) in healthy adults aged between 18 and 65 years in China |
| Objectives | Primary objectives: safety and tolerability of RSAV in healthy adults |
| Secondary objectives: immunogenicity of RSAV in health adults | |
| Target population | Healthy adults aged between 18 and 65 years |
| Sample size | 144 participants |
| Rational and background | Staphylococcus aureus (S. aureus), a gram positive bacteria, is one of a major cause of hospital-associated and community-associated infections worldwide. These infections range from minor skin and soft tissue infections to the major life-threatening invasive infections (1). The frequencies of both community-acquired and hospital-acquired staphylococcal infections have increased steadily in recent years. Treatment of these infections caused by S. aureus has become more difficult because of the emergence of multidrug-resistant strains (MRSA) (4). |
| A comprehensive survey conducted in clinical sites in the USA, Canada, Europe, found that S. aureus accounted for 22% of all blood infections (8,929 of 40,497 infections), 23.2% of all lower respiratory tract (3,371/14,552 infections) and 39.2% (2,928/7,474 infections) of all skin and soft tissue infections (2). In China, S. aureus was the most common clinical gram-positive bacteria with the highest isolated rate, and the MRSA accounted for 44.6% of all the isolated strains (3). It is critical to develop an effective vaccine to control the infection caused by S. aureus and reduce the usage of antibiotics in order to decrease the drug resistance of S. aureus | |
| Currently, there is no effective vaccine against S. aureus has been reported. Early clinical trials of several S. aureus vaccine candidates such as Nabi’s StaphVax and PentaStaph, Intercell’s/Merck’s V710, VRi’s SA75, were all found to be invalid against infectious disease caused by S. aureus (7). Although some vaccine candidates were highly |
|
| The experimental vaccine contains five antigens: the alpha hemolysin (Hla), iron-regulated surface determinant B (IsdB), S. aureus protein A (SpA), enterotoxin B (SEB), manganese transport protein C (MntC). In preclinical studies of this vaccine candidate, a total of 16,000 mice and 40 cynomolgus monkeys were tested. The vaccine was safe and immunogenic, showing a protection of over 85% | |
| This is a phase 1 trial to evaluate the safety and tolerability and preliminary immunogenicity (including both humoral and cellular immune responses) of this novel RSAV in healthy adults aged between 18 and 65 years | |
| Immunization schedule | Immunization schedule: four shots of intramuscular injection at day 0, day 3, day 7, and day 14, respectively |
| Trial design | This is a single-center, randomized, double-blind, placebo-controlled clinical trial |
| A total of 144 participants will be included and randomly assigned to the low-dose group, middle-dose group, |
|
| Randomization and blind | |
| A two-stage randomization method will be applied in this study. In the first stage randomization, a total of 144 |
|
| Primary endpoints | Occurrence of solicited adverse reactions within 21 days after the first vaccination |
| Secondary endpoints | Safety: |
| Abnormal change of safety laboratory measures (routine blood test and blood biochemical test) on day 3, day 7, day 10, day 14, and day 17 compared with the baseline | |
| Occurrence of unsolicited adverse reactions within 42 days after the first vaccination | |
| Occurrence of serious adverse events (SAEs) during the whole follow-up period (6 months) | |
| Immunogenicity: | |
| Antibody geometric mean titers (GMTs) of antigen-specific antibodies in serum on day 7, day 14, day 21, day 42, month 3, and month 6 post-vaccination | |
| The geometric mean fold increase (GMFI) of antigen-specific antibodies in serum on day 7, day 14, day 21, day 42, month 3, and month 6 post-vaccination | |
| Seroconversion rate of antigen-specific antibodies in serum on day 7, day 14, day 21, day 42, month 3, and month 6 post-vaccination | |
| Exploratory endpoints | S. aureus colonization: |
| The nasal S. aureus colonization in participant’s nose on day 3, day 7, day 10, day 14, day 17, day 21, day 42, month 3, and month 6 post-vaccination | |
| Functional antibody response: | |
| Functional antibody titres on day 7, day 14, day 21, day 42, month 3, and month 6 post-vaccination | |
| Cellular immune response: | |
| Cellular immune responses on day 7, day 14, day 21, day 42, month 3, month 6 post-vaccination | |
| Scheduled site visits | Visit 0 (within 7 days before vaccination): informed consent, exclusion criteria screening, physical examination, blood sample taking for baseline laboratory measures |
| Visit 1 (day 0): exclusion criteria screening, randomization nose swab, and blood sample collection, vaccination, safety observation | |
| Visit 2 (day 3): exclusion criteria screening, nose swab and blood sample collection, vaccination, safety observation | |
| Visit 3 (day 7): exclusion criteria screening, nose swab and blood sample collection, vaccination, safety observation | |
| Visit 4 (day10): nose swab and blood collection, safety observation | |
| Visit 5 (day 14): exclusion criteria screening, nose swab and blood sample collection, vaccination, safety observation | |
| Visit 6 (day 17): nose swab and blood collection, safety observation | |
| Visit 7 (day 21): nose swab and blood collection, safety observation | |
| Visit 8 (day 42): nose swab and blood collection, safety observation | |
| Visit 9 (month 3): nose swab and blood collection, safety observation | |
| Visit 10 (month 6): nose swab and blood collection, safety observation | |
| Inclusion criteria | Healthy volunteers aged between 18 and 65 years |
| Willing to comply with the clinical trial protocol and complete all the visits planed during the study period | |
| Able to understand the content of informed consent and willing to sign the informed consent | |
| Able to complete the diary card independently | |
| For females only (18–49 years), a negative urine pregnancy test and willing to practice continuous effective contraception during the study | |
| Axillary temperature ≤37.0 °C | |
| Exclusion criteria | First immunization exclusion standard: |
| Prior receipt of S. aureus vaccine | |
| Any confirmed S. aureus infection disease in the past 12 months | |
| History of asthma, hereditary angioneurotic edema, diabetes, malignancy or other serious disease. Participation in the clinical trial is likely to increase the disease risk and interfere with the observation of clinical trial index | |
| Prior blood donation or Blood loss over 400 mL in the last 3 months | |
| Coagulation disorders (coagulation factor deficiency, coagulopathy or platelet disorder) diagnosed by doctors, or obvious bruises or blood coagulation noticed. | |
| History of allergic disease likely to be exacerbated by any component of the vaccine, including allergy, urticarial, respiratory difficulty, angioneurotic edema or abdominal pain | |
| Any autoimmune disease or immunodeficient state, parents, brother and sister with autoimmune disease or immunodeficient disease | |
| Taking immunoglobulins and/or any blood products within the last 12 months | |
| Asplenia, functional asplenia or asplenia caused by any situation or splenectomy | |
| Any acute disease or acute attack of chronic disease in last 7 days | |
| History of thyroidectomy or thyroid disease requiring treatment in the last 12 months | |
| Immunosuppressant, cytotoxic therapy, inhaled corticosteroid (excluding corticosteroids spray treatment of allergic rhinitis, acute and non-concurrent corticosteroids treatment) | |
| Participation in another research study involving receipt of an investigational product in the last 30 days | |
| Woman who is breast-feeding | |
| Prior administration of attenuated vaccine in last 28 days | |
| Prior administration of subunit vaccine, inactivated vaccine or allergic therapy in last 14 days | |
| Current anti-tuberculosis prophylaxis or therapy | |
| Any other conditions may compromise the safety or availability of participants in the judgment of the investigator | |
| Following immunization exclusion standard: | |
| Any grade 3 or more serious adverse reaction happen since the last vaccination | |
| Other condition violates the inclusion criteria or meets the exclusion criteria is noticed after the first immunization | |
| Acute or chronic infections at the vaccination day (axillary temperature >37.0 °C) | |
| According to the investigator, the participant should not continue participating in the study | |
| Role of the sponsor | Sponsors participate in the trial design, but will not participate in other process of the trial, including data collection, statistical analysis, data interpretation, and drafting study report |
| Criteria for pausing or early termination | On the first two days of enrollment, no more than 12 participants will be recruited and vaccinated each day. And no more than 24 participants will be recruited and vaccinated in each day in the following recruiting period |
| Data and Safety Monitoring Board (DSMB) will review the reported safety data during the study period. If an increase of risk for participants is noticed, the DSMB should promptly inform the principle investigator and sponsors. Sponsors, investigators and DSMB will have a panel meeting, and make a final decision whether need to pause or call an early termination of the study | |
| Administration of study injections and new enrollments will be paused, if: | |
| One serious adverse event may be associated with vaccination, or | |
| Occurrence of grade 3 adverse reaction may be associated with vaccination in 10% of participants or more (including injection-site reaction, systemic reaction, and change of the safety laboratory measures) | |
| The study may come to an early termination, if: | |
| One vaccination-associated serious adverse event, or | |
| Occurrence of grade 3 adverse events associated with vaccination in 15% of participants or more (including injection-site reaction, systemic reaction, and change of the safety laboratory measures), or | |
| Required by sponsor, or | |
| Required by regulatory authority, or | |
| Required by institutional review board (IRB) | |
| Interim analyses | Interim analyses will be conducted by an independent statistical party after all the participant complete visit 8 on day 42. The data collected from day 0 to day 42 will be unblinded by representatives of statistical party, sponsor, clinical research investigator and Clinical Research Organization (CRO). The un-blinding data will not be disclosed to the participants, staff who perform the site works and follow-up, and the laboratory staff who test the bio-samples. Therefore, the clinical trial site will still be kept in blind. The interim results will not influence the later safety or immunogenicity endpoint assessments |
| Final analyses | Final analyses will be conducted by the statistical party, after the last participant completes visit 10 on 6 month, and all the safety data and immunogenicity data collected after day 42 up to month 6 will be reviewed and then locked for the final statistical analysis |
| Table of Content | ||
|---|---|---|
| DOCUMENT HISTORY | 2 | |
| CLINICAL TRIAL CONTACT LIST | 4 | |
| PROTOCOL SUMMARY | 6 | |
| ABBREVIATIONS | 14 | |
| 1. | OBJECTIVES AND BRIEF INTRODUCTION | 16 |
| 2. | STUDY SITE | 16 |
| 3. | BACKGROUND AND RATIONALE | 16 |
| 3.1. | DISEASE BACKGROUND | 16 |
| 3.2. | PATHOGENY CHARACTERISTICS | 17 |
| 3.2.1. | S. aureus typing | 17 |
| 3.2.2. | Epidemiological character | 17 |
| 3.3. | VACCINE DEVELOPMENT IN OTHER COUNTRIES | 18 |
| 3.4. | INTRODUCTION OF THE EXPERIMENTAL VACCINES | 19 |
| 3.5. | BENEFITS AND RISKS | 20 |
| 4. | STUDY OBJECTIVES | 20 |
| 4.1. | PRIMARY OBJECTIVE | 20 |
| 4.2. | SECONDARY OBJECTIVE | 20 |
| 5. | STUDY DESIGN | 21 |
| 5.1. | STUDY DESIGN AND METHOD DESCRIPTION | 21 |
| 5.1.1. | Blinding | 21 |
| 5.1.2. | Method of unblinding and breaking the study blind | 21 |
| 5.2. | STUDY ENDPOINTS | 22 |
| 5.2.1. | Primary endpoints | 22 |
| 5.2.2. | Secondary endpoints | 22 |
| 5.2.3. | Exploratory endpoints | 23 |
| 5.3. | STUDY PROCEDURES | 23 |
| 5.4. | SAMPLE SIZE | 25 |
| 5.5. | CRITERIA FOR PAUSING THE STUDY OR AN EARLY TERMINATION | 25 |
| 5.6. | DURATION OF STUDY | 26 |
| 6. | PARTICIPANTS | 26 |
| 6.1. | PARTICIPANTS SELECTION | 26 |
| 6.2. | INCLUSION CRITERIA | 26 |
| 6.3. | EXCLUSION CRITERIA | 26 |
| 6.4. | WITHDRAW FROM THE STUDY | 27 |
| 7. | INVESTIGATIONAL PRODUCT | 28 |
| 7.1. | INVESTIGATIONAL VACCINE | 28 |
| 7.2. | ADMINISTRATION | 28 |
| 7.3. | TRANSPORTATION AND STORAGE | 29 |
| 8. | SAFETY ASSESSMENTS AND ADVERSE EVENT REPORTING | 29 |
| 8.1. | ADVERSE EVENT AND ADVERSE REACTION | 29 |
| 8.2. | GRADING FOR ADVERSE EVENTS | 30 |
| 8.2.1. | Injection site | 30 |
| 8.2.2. | Systemic reactions | 31 |
| 8.2.3. | Safety laboratory measures | 32 |
| 8.3. | SERIOUS ADVERSE EVENT/REACTION (SAE) | 33 |
| 8.3.1. | Reporting serious adverse events (SAEs) | 34 |
| 8.3.2. | Expedited reporting of SUSARs to regulatory authorities and investigators | 34 |
| 8.4. | SAFETY DATA MONITORING BY DSMB | 35 |
| 8.5. | WITHDRAWAL DUE TO ADVERSE EVENTS (SEE ALSO SECTION ON PARTICIPANT WITHDRAWAL) | 36 |
| 9. | LABORATORY ASSAY AND IMMUNOGENICITY ASSESSMENTS | 36 |
| 9.1. | ANTIGEN-SPECIFIC ANTIBODY RESPONSES | 36 |
| 9.2. | CELLULAR IMMUNITY RESPONSES | 36 |
| 9.3. | FUNCTIONAL ANTIBODY LEVEL | 36 |
| 9.4. | NASAL SAMPLING SWAB | 36 |
| 10. | DATA COLLECTION AND MANAGEMENT | 37 |
| 10.1. | SOURCE DOCUMENTS AND SOURCE DATA | 37 |
| 10.2. | CLINICAL DATA MANAGEMENT | 37 |
| 10.3. | SAE DATA MANAGEMENT | 38 |
| 10.4. | ARCHIVING | 38 |
| 10.5. | DATABASE CREATION AND DATA ENTRY | 39 |
| 10.6. | THE DATABASE LOCK | 39 |
| 11. | STATISTICS PLAN AND STATISTICAL ANALYSIS | 39 |
| 11.1. | INTERIM STATISTICAL ANALYSIS | 39 |
| 11.2. | THE FINAL ANALYSIS | 40 |
| 11.3. | ANALYZED DATA SETS DEFINITION | 40 |
| 11.4. | STATISTICAL METHODS | 40 |
| 12. | ETHICAL AND LEGAL ISSUES | 41 |
| 12.1. | GUIDELINE | 41 |
| 12.2. | INSTITUTIONAL REVIEW BOARD | 41 |
| 12.3. | ETHICAL CONDUCT OF THE STUDY | 42 |
| 12.4. | PROTOCOL AMENDMENTS AND ADMINISTRATIVE CHANGES | 42 |
| 12.5. | CONFIDENTIALITY OF DATA AND ACCESS TO PARTICIPANT RECORDS | 43 |
| 13. | FINANCIAL CONTRACT AND INSURANCE COVERAGE | 43 |
| 14. | PUBLICATION OF STUDY RESULTS | 43 |
| 15. | REFERENCE | 43 |
| AE | Adverse Event | NIAID | The National Institute of Allergy and Infectious Diseases | |
|---|---|---|---|---|
| AR | Adverse Reaction | ORFs | open reading frames | |
| ATP | According to Protocol | PVL | Leukocidin | |
| CI | Confidence Interval | PBMC | Peripheral blood mononuclear cells | |
| CRF | Case Report Form | PPS | per-protocol set | |
| CRO | Clinical Research Organization | IRB | institutional review board | |
| CFDA | China Food and Drug Administration | ITT | Intent-to-treat | |
| DSMB | Data and Safety Monitoring Board | IEC | Independent Ethics Committee | |
| ELISA | Enzyme-linked immune sorbent assay | IsdB | Iron Surface Determinant B | |
| ELISpot | Enzyme-linked immune spot assay | RSAV | the recombinant staphylococcus aureus vaccine | |
| FAS | Full Analysis Set | S. aureus | Staphylococcus aureus | |
| GCP | Good Clinical Practice | StrA | Sortase A | |
| GMT | Geometric Mean Titer | SE | staphylococcus aureus enterotoxin | |
| GMFI | Geometric Mean Fold Increase | SAEs | Serious Adverse Events | |
| GSK | Glaxo Smith Kline | SOP | Standard Operation Procedure | |
| Hla | Alpha hemolysin | SS | Safety set | |
| MedDRA | Medical Dictionary for Regulatory Activities | SpA | Staphylococcal Protein A | |
| MRSA | methicillin - resistant Staphylococcus aureus | SFDA | state Food and Drug Administration | |
| MSCRAMMs | microbial surface components recognizing adhesive matrix molecules | WHO | The World Health Organization | |
| mSEB | Staphylococcal Enterotoxin B mutant | TSST 1 | toxic shock toxin-1 | |
| MntC | manganese ion binding protein C | |||
| NIFDC | National institute for food and drug control |
Table S1
| Vaccine | Antigen target | Company | Clinical trial phase |
|---|---|---|---|
| V710 | Iron surface determinant B (IsdB) | Merck/Intercell | Phase 3 |
| StaphVax | CPS types5+8 | Nabi Biota pharmaceuticals | Phase 3 |
| Bivalent rLukS-PV/rAT | rLukS-PV/rAT | Nabi Biota pharmaceuticals | Phase 2 |
| SA3Ag | CP5\CP8\ClfA | Pfizer | Phase 2 |
| SA4Ag | CP5\CP8\ClfA\MntC | Pfizer | Phase 2 |
| NDV-3 | Candida Als3 | Novartis | Phase 2 |
| GSK2392103A | Recombinant proteins | GSK | Phase 1 |
| GSK2392105A | |||
| GSK2392106A | |||
| GSK2392019A | |||
| STEBVax | Staphylococcal enterotoxin B | NIAID | Phase 1 |
Table S2
| Visit (V) | Scheduled time point for visit | Time window |
|---|---|---|
| Visit 0 (V0) | Within 7 days before vaccination | – |
| Visit 1 (V1) | The first vaccination at day 0 | – |
| Visit 2 (V2) | Day 3 after the first vaccination | +1 day |
| Visit 3 (V3) | Day 7 after the first vaccination | +1 day |
| Visit 4 (V4) | Day 10 after the first vaccination (3 days after V3) | ±1 day |
| Visit 5 (V5) | Day 14 after the first vaccination | ±1 day |
| Visit 6 (V6) | Day 17 after the first vaccination (3 days after V5) | ±1 day |
| Visit 7 (V7) | Day 21 after the first vaccination (7 days after V5) | +2 days |
| Visit 8 (V8) | Day 42 after the first vaccination (28 days after V5) | +3 days |
| Visit 9 (V9) | Month 3 after the first vaccination | ±7 days |
| Visit 10 (V10) | Month 6 after the first vaccination | ±7 days |
Table S3
| Activities | V0 | V1 | V2 | V3 | V4 | V5 | V6 | V7 | V8 | V9 | V10 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Within 7 days before day 0 | D0 | D3 | D7 | D10 | D14 | D17 | D21 | D42 | M3 | M6 | |
| Informed consent | ☆ | ||||||||||
| Collecting demographic information | ☆ | ||||||||||
| Preliminarily check inclusion and exclusion criteria | ☆ | ||||||||||
| Physical examination: | |||||||||||
| Height, weight, BMI | ☆ | ||||||||||
| Respiratory rate, pulse rate, blood pressure | ☆ | ||||||||||
| Urine pregnancy test (female) | ☆ | ||||||||||
| Routine blood test | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |||||
| Blood biochemical test | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |||||
| Pre-vaccination body temperature | ☆ | ☆ | ☆ | ☆ | |||||||
| Inclusion and exclusion criteria before enrolled | ☆ | ||||||||||
| Follow-up vaccination exclusion criteria | ☆ | ☆ | ☆ | ||||||||
| Randomization | ☆ | ||||||||||
| Serum analyses: | |||||||||||
| Five antigen-specific antibody response by ELISA | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ||||
| Functional antibody level | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ||||
| Cellular immune responses | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ||||
| Nasal sampling swab | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |
| Vaccination | ☆ | ☆ | ☆ | ☆ | |||||||
| Observation for 30 minutes post-vaccination, participates safety observation training | ☆ | ☆ | ☆ | ☆ | |||||||
| Distribution of diary cards, ruler, signing-pen, thermometer | ☆ | ☆ | ☆ | ||||||||
| Record on the “Vaccination and visits record” | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |
| Participates safety observations self-report in diary cards | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |||
| Report serious adverse events | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | ☆ | |
| Return of diary cards | ☆ | ☆ | ☆ |
Table S4
| Activities | V0 | V1 | V2 | V3 | V4 | V5 | V6 | V7 | V8 | V9 | V10 |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Within 7 days before day 0 | Day0 | Day3 | Day7 | Day10 | Day14 | Day17 | Day21 | Day42 | Mon 3 | Mon6 | |
| Clinical evaluations | |||||||||||
| Routine blood test | 2 | – | 2 | 2 | 2 | 2 | 2 | – | – | – | – |
| Blood biochemical test | 5 | – | 5 | 5 | 5 | 5 | 5 | – | – | – | – |
| Immunogenicity | |||||||||||
| Antibody assays and serum storage | – | 8 | – | 8 | 8 | 8 | 8 | 8 | 8 | ||
| PBMC and plasma for storage | – | 16 | – | 16 | 16 | 16 | 16 | 16 | 16 | ||
| Daily volume | 7 | 24 | 7 | 31 | 7 | 31 | 7 | 24 | 24 | 24 | 24 |
| Cumulative volume | 7 | 31 | 38 | 69 | 76 | 107 | 114 | 138 | 162 | 186 | 210 |
Table S5
| Laboratory indexes | Mild (grade 1) | Moderate (grade 2) | Severe (grade 3) | Potentially life threatening (grade 4) |
|---|---|---|---|---|
| Hemoglobin (female) change from baseline value, gm/dL | Any increase –1.5 | 1.6–2.0 | 2.1–5.0 | >5.0 |
| Hemoglobin (male) change from baseline value, gm/dL | Any increase –1.5 | 1.6–2.0 | 2.1–5.0 | >5.0 |
| Hemoglobin (female), gm/dL | 12.0–13.0 | 10.0–11.9 | 8.0–9.9 | <8.0 |
| Hemoglobin (male), gm/dL | 12.5–14.5 | 10.5–12.4 | 8.5–10.4 | <8.5 |
| WBC increase, cell/mm3 | >13,000/mm3 (>13×109/L) | 13,000–15,000/mm3 (13–15×109/L) | 15,000–30,000/mm3 (15–30×109/L) | >30,000/mm3 (>30×109/L) |
| WBC decrease, cell/mm3 | 2,500–3,500/mm3 (2.5–3.5×109/L) | 1,500–2,499/mm3 (1.5–2.499×109/L) | 1,000–1,499/mm3 (1.0–1.499×109/L) | <1,000/mm3 |
| Liver function tests—ALT, AST increase by factor | 1.25–2.5× ULN* | 2.6–5× ULN | 5.1–10× ULN | >10× ULN |
| Creatinine increase by factor | 1.1–1.5× ULN | 1.6–3.0× ULN | 3.1–6× ULN | >6× ULN |
*ULN, is the upper limit of the normal range.
Table S6
| Name | Title | Work units | Major | Position |
|---|---|---|---|---|
| Jie-Lai Xia | Professor | Public Health Department, Fourth Military Medical University | Statistics | Chairman |
| Xuan-Yi Wang | Professor | Institute of Biomedical Sciences Fudan University | Epidemiology | Member |
| Tao Wang | Professor | Institute of immunology, Second Military Medical University | Immunology | Member |
| Xiang-Dong Zhou | Professor of Medicine | Southwest Hospital | Clinical infection disease | Member |
| Hui-Wang | Professor of Medicine | Peking People’s Hospital | Clinical infection disease | Member |
Acknowledgments
Funding: None.
Footnote
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/jphe.2016.12.11). FZ serves as an Deputy Editor-in-Chief of Journal of Public Health and Emergency from Jan 2017 to Dec 2022. The other authors have no conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved. The study was conducted in accordance with the Declaration of Helsinki (as revised in 2013). The trial was approved by the Ethics Committee of Jiangsu Provincial Center for Disease Prevention and Control. Written informed consent was obtained from each participant before any study-related activity began.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Adhikari RP, Kort T, Shulenin S, et al. Antibodies to S. aureus LukS-PV Attenuated Subunit Vaccine Neutralize a Broad Spectrum of Canonical and Non-Canonical Bicomponent Leukotoxin Pairs. PLoS One 2015;10:e0137874 [Crossref] [PubMed]
- Diekema DJ, Pfaller MA, Schmitz FJ, et al. Survey of infections due to Staphylococcus species: frequency of occurrence and antimicrobial susceptibility of isolates collected in the United States, Canada, Latin America, Europe, and the Western Pacific region for the SENTRY Antimicrobial Surveillance Program, 1997-1999. Clin Infect Dis 2001;32:S114-32. [Crossref] [PubMed]
- Hu F, Zhu D, Wang F, et al. 2014 CHINET bacterial drug resistance surveillance in China. Chinese journal of infection and chemotherapy 2015:401-10.
- Blot S, Vandewoude K, Colardyn F. Staphylococcus aureus infections. N Engl J Med 1998;339:2025-6; author reply 2026-7. [Crossref] [PubMed]
- Fattom AI, Horwith G, Fuller S, et al. Development of StaphVAX, a polysaccharide conjugate vaccine against S. aureus infection: from the lab bench to phase III clinical trials. Vaccine 2004;22:880-7. [Crossref] [PubMed]
- Moustafa M, Aronoff GR, Chandran C, et al. Phase IIa study of the immunogenicity and safety of the novel Staphylococcus aureus vaccine V710 in adults with end-stage renal disease receiving hemodialysis. Clin Vaccine Immunol 2012;19:1509-16. [Crossref] [PubMed]
- Tacconelli E. Methicillin-resistant Staphylococcus aureus: source control and surveillance organization. Clin Microbiol Infect 2009;15:31-8. [Crossref] [PubMed]
- The registration and management of drugs. Chinese journal of new drugs and clinical 2008;27:303-12. Available online: http://www.sda.gov.cn/WS01/CL0053/24529.html
- Allen KB, Fowler VG Jr, Gammie JS, et al. Staphylococcus aureus Infections After Elective Cardiothoracic Surgery: Observations From an International Randomized Placebo-Controlled Trial of an Investigational S aureus Vaccine. Open Forum Infect Dis 2014;1:ofu071 [Crossref] [PubMed]
- Daum RS, Spellberg B. Progress toward a Staphylococcus aureus vaccine. Clin Infect Dis 2012;54:560-7. [Crossref] [PubMed]
- Aboud-Pirak E, Lubin I, Pirak ME, et al. Lethally irradiated normal strains of mice radioprotected with SCID bone marrow develop sensitivity to low doses of staphylococcal enterotoxin B. Immunol Lett 1995;46:9-14. [Crossref] [PubMed]
- Anderson AS, Scully IL, Timofeyeva Y, et al. Staphylococcus aureus manganese transport protein C is a highly conserved cell surface protein that elicits protective immunity against S. aureus and Staphylococcus epidermidis. J Infect Dis 2012;205:1688-96. [Crossref] [PubMed]
- Kuklin NA, Clark DJ, Secore S, et al. A novel Staphylococcus aureus vaccine: iron surface determinant B induces rapid antibody responses in rhesus macaques and specific increased survival in a murine S. aureus sepsis model. Infect Immun 2006;74:2215-23. [Crossref] [PubMed]
- Missiakas D, Schneewind O. Staphylococcus aureus vaccines: Deviating from the carol. J Exp Med 2016;213:1645-53. [Crossref] [PubMed]
Cite this article as: Zhu YY, Li JX, Zeng H, Zeng M, Meng FY, Zou QM, Zhu FC. Safety, tolerability, and preliminary immunogenicity of a recombinant Staphylococcus aureus vaccine (RSAV) in healthy adults between 18 and 65 years in China: study protocol for a randomized, double-blind, placebo-controlled, phase 1 trial. J Public Health Emerg 2017;1:25.